Promising targets for deafness drug discovery
Posted: 23 November 2020 | Hannah Balfour (Drug Target Review) | No comments yet
In this article, Janssen Pharmaceuticals’ Global Head of Discovery Neuroscience discusses the findings of their recent study which highlighted new roles for two gene products involved in human deafness which could enable drug discovery for some prevalent auditory disorders.

“Of our five senses, the auditory system is unique in that it is the only one to receive feedback from the brain,” said David Bredt, MD, PhD, Global Head of Discovery Neuroscience at Janssen Pharmaceuticals. However, he noted that age-related hearing loss is growing in prevalence with louder noises and people listening to music through earbuds. “By some estimates, it is the fourth greatest disease burden by costs. The hair cells in the ear cannot regenerate, so once these die people lose their hearing. Hearing aids are the only treatment currently available for age-related hearing loss; they are helpful in that they allow people to hear sounds but are very unsatisfactory in terms of sound discrimination. Damage to hair cells can also cause people to develop a very unfortunate and untreatable condition called tinnitus – ringing in the ears – the magnitude of which is really underappreciated.”
To address this, Bredt and his colleagues expressed a target receptor in a cell model and used genome-wide screening to establish which factors are required for its assembly, expression and signalling. The team believes that by better understanding the function of this receptor, they may be able to develop therapeutics for various prevalent auditory disorders.
Why target the α9α10 receptor when exploring auditory disorders?
Efferent medial olivocochlear (MOC) neurons, which carry signals from the brain to the ear, synapse onto the outer hair cells of the cochlea.1 The cochlea is the hollow, spiral-shaped bone in the inner ear responsible for transducing sound waves into electrical signals that can be interpreted by the brain through a process called mechanotransduction.2 There are two types of hair cells found in the cochlea: inner hair cells, which create afferent electrical impulses via mechanotransduction and outer ear cells, which increase the amplitude of sound waves in the fluid (called endolymph) surrounding the cochlea.2 Increasing the amplitude of sound waves is the same as increasing their volume – making it more likely that a sound wave will be transduced by the inner ear cells.
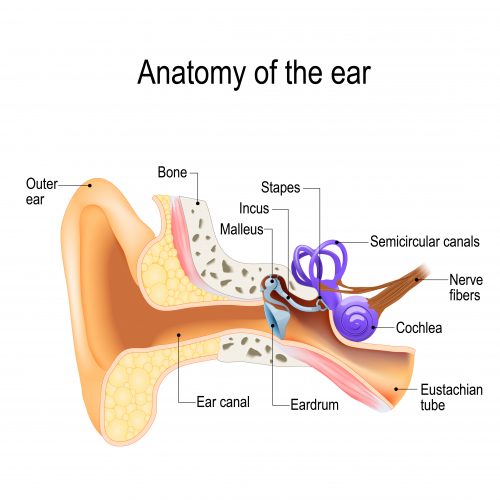
Impulses from the efferent MOC neurons decrease the ability of outer ear cells to enhance the amplitude of the sound waves. Inner ear cells are particularly sensitive to loud noises and can be irreparably damaged or destroyed if exposed to sounds of 85 decibels or greater for an eight-hour period, resulting in permanent hearing loss.2 Therefore, the efferent signals from MOC neurons protect against auditory trauma. They also allow humans to selectively focus on particular auditory inputs and improve the signal-to-noise ratio, enabling us to discriminate between individual sounds and background noise. An example of this is focusing on a single conversation occurring in a noisy room.
Studies have shown that a type of nicotinic acetylcholine receptor (nAChR), called the α9α10 receptor, is located in the synapse between efferent MOC neurons and outer hair cells. When activated, MOC neurons release acetylcholine into the synaptic junction, activating the α9α10 nAChRs on outer hair cells. These receptors couple to SK2 potassium channels and cause hyperpolarisation of the hair cells, suppressing their motility and inhibiting their ability to enhance sound wave amplitude.3
Studies in mice have shown that mutations to either the α9 or α10 subunits of the receptor have profound auditory effects. Such effects, which are highlighted in Janssen’s new study,4 include:
- disrupting either α9 or α10 abolishes acetylcholine signalling in both outer and inner hair cells, resulting in impaired selective attention
- mice lacking α9 develop cochlear abnormalities
- gain-of-function mutations to α9 protect mice from acoustic trauma.
This evidence led Bredt to suggest that the α9α10 receptor could help prevent sound-induced hearing loss and potentially aid in the repair and rescue of damaged hair cells before they die. “Upregulating its protective effects could therefore have a variety of applications in both industrial and military settings,” he said.
Difficulties in studying the α9α10 nAChR
Despite the α9α10 receptor being a potentially highly selective (as its endogenous activity has only been recorded in cochlear and vestibular ear cells) and desirable target, Bredt explained that studying it for drug development is challenging: “For drug discovery, we typically want to express the target receptors in cell lines and then use robots to screen as many compounds as possible. This particular receptor was never amenable to that because it does not function in recombinant cell lines. One of the major breakthroughs in this new study is that, for the first time, it allows drug discovery on this very attractive, but previously elusive, receptor.”
Findings of the study
Functional partners of the α9α10 receptor
While it has been established that the α9α10 nAChR is functionally expressed on outer hair cells after the onset of hearing and persists into adulthood, what accessory proteins enable its assembly and function were unknown. In the study, researchers used genome-wide complimentary DNA (cDNA) screening to identify functional partners of the α9α10 receptor.

It was found that two products of genes implicated in the development of human non-syndromic deafness, transmembrane inner ear (TMIE) and TMEM132e, were necessary for the α9α10 receptor to function. Co-expression of either TMIE or TMEM132e was sufficient for the nAChR to reconstitute in cells and produce small responses to acetylcholine. When choline acetyltransferase (ChAT) was expressed with TMIE in the cells, they were found to have a pharmacological profile similar to in vivo outer hair cells and had a large desensitisation response to acetylcholine. The team concluded from their experiments that ChAT and the nicotinic ligands it produces enable α9α10 receptor assembly, stabilisation and surface expression and that co-expression of a transmembrane protein, eg, TMIE or TMEM132e, is required for ion channel activity.
TMIE and TMEM132e mutations in non-syndromic deafness
TMIE and TMEM132e are known to be enriched in the tips of inner hair cells and involved in mechanotransduction. Mutations in TMEM132e are associated with autosomal‐recessive non-syndromic hearing loss. The TMIE protein was shown in the study to localise to both the tip of the hair cell stereocilia, where it is an essential component of inner hair cell mechanotransduction channel machinery and in the cell body. The latter is a new finding which suggests the protein might have other, currently unidentified roles in the auditory system. Mice with TMIE protein mutations were also shown to have enhanced inner hair cell α9α10 receptor activity at development stages where MOC innervation typically would have stopped (postnatal day eight), resulting in deafness.
The α9α10 nAChR in other disorders
Future therapeutics targeting either the receptor or its functional partners could be used to protect against or restore hearing loss due to acoustic trauma”
Bredt explained that although auditory disorders are not a focus for Janssen, they are particularly interested in the role of the α9α10 receptor in the immune system. “There is some strong evidence that it is involved in neuropathic pain and neuroinflammatory disorders,” he said. “There is data that inhibitors of this receptor are helpful in treating chronic pain and multiple sclerosis. So far, the data has been collected from animal models, because researchers were unable to functionally express it in cell lines; however, with the techniques and factors identified in this study, it should now be easier to explore.”
He added that they are also looking at this receptor and others in neurodegenerative disorders, such as Alzheimer’s and Parkinson’s disease, which are associated with dysregulation of the neuro-immune axis.
Conclusion
In their study, the researchers were able to identify which factors are essential for the α9α10 receptor to be functionally expressed in in vitro cell lines for efficient drug discovery. Future therapeutics targeting either the receptor or its functional partners could be used to protect against or restore hearing loss due to acoustic trauma in the inner ear. During the study they also found that two of these factors were gene products associated with non-syndromic deafness and that they may have previously undiscovered roles in the auditory system.
The study concluded that being able to functionally reconstitute α9α10 receptors has important biochemical and biomedical implications, including enabling high-throughput screening and identification of pharmacological agents to modulate α9α10 receptor function for auditory disorders.
References
- Smith, D.W. and Keil, A. The biological role of the medial olivocochlear efferents in hearing: separating evolved function from exaptation. Syst. Neurosci., 25 February 2015; https://doi.org/10.3389/fnsys.2015.00012
- Casale, J., Kandle, P.F., Murray, I., et al. Physiology, Cochlear Function [Internet]. StatPearls. 2020 January [Updated 2020 Aug 24]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK531483/
- Dallos, P., He, D.Z.Z., Lin, X., et al. Acetylcholine, Outer Hair Cell Electromotility, and the Cochlear Amplifier. Journal of Neuroscience, 15 March 1997, 17 (6) 2212-2226; https://doi.org/10.1523/JNEUROSCI.17-06-02212.1997
- Gu, S., Knowland, D., Matta, J.A., et al. Hair cell α9α10 nicotinic acetylcholine receptor functional expression regulated by ligand binding and deafness gene products. Proceedings of the National Academy of Sciences, September 2020, 117 (39) 24534-24544; https://doi.org/10.1073/pnas.2013762117
Related topics
Disease Research, Drug Targets, In Vitro, Neurosciences, Protein
Related conditions
Auditory disorders, Non-syndromic deafness
Related organisations
Janssen Pharmaceuticals
Related people
David Bredt MD PhD