Scientists discover bacteria’s clever defence mechanism
Posted: 19 November 2014 | Diamond
Structure of EzrA protein could help identify new antibiotic targets…
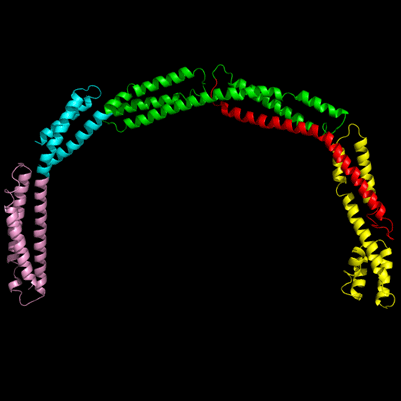
A close-up of the EzrA protein, depicted as a ribbon
Scientists have uncovered important new information about how bacteria grow and multiply, potentially leading to the discovery of much-needed new antibiotic drugs. Using Diamond Light Source – the UK’s synchrotron science facility – scientists have determined the structure of EzrA: a protein that helps bacteria to build their defensive cell walls as they divide and spread.
All cells have an outer layer that forms a protective barrier around the important genetic information within. For some bacteria, a strong outer wall helps the invading cells to survive attack as they spread through the body; it is this wall that is the target of many commonly used antibiotics. However, there are a number of holes in our knowledge of how the wall develops as bacterial cells divide and spread throughout the body.
Bacteria spread through cell division; once a bacterial cell has grown to its full size, it starts to tighten a belt around its middle, and the belt grows ever tighter until it eventually severs the old cell into two separate cells. As this process is taking place, the cell has to make an exact copy of its genome, so that each new bacterial cell contains the same genetic information as all the others. In this way, bacteria ensure that they maintain their powerful defence mechanisms in each new cell as they spread through the body.
But a team from the University of Newcastle have now used the powerful X-ray light on Diamond’s I04-1 beamline to identify the atomic structure of the protein that helps to drive this system of divide and conquer. EzrA is an important protein for regulating the process of cell division. It helps to control another protein, FtsZ, which manages the tightening of the belt that causes the bacterial cell to divide in two. Whilst the atomic make-up of FtsZ was already understood, advanced synchrotron techniques have now allowed scientists to go one step deeper and uncover EzrA, the protein that supports the process.
The work, published in Nature Communications, reveals that the atomic structure of the EzrA protein is as complex as it is beautiful. An unusual conformation, EzrA is shaped like a hoop, the middle of which is able to accommodate the belt-tightening protein, FtsZ. EzrA’s hoop-like shape prevents the FtsZ protein from escaping until it has fulfilled its purpose: causing the cell belt to contract and divide the cell in two whilst the genome of the parental cell is being copied and passed to the daughter.
The discovery of the EzrA structure is particularly significant because it may help to identify new targets for future antibiotics. Understanding the atomic structure of EzrA could help scientists to develop ways of disabling it, thus preventing bacteria from dividing correctly. Without this vital protein, bacteria would fail to create a protective cell wall for each new cell created as it divides and spreads, leaving new cells open to attack.
Rick Lewis, Professor of Structural Biology at Newcastle and team leader on the project, comments: “The structure of Ezra is the most beautiful and intriguing that I have ever worked on; if someone had told me four years ago when the work commenced in earnest that EzrA formed this structure, I would not have believed them! Nature continues to surprise and to inspire. I hope that the amazing work carried out by my group, and that of our fantastic colleagues and collaborators, will inspire others to delve deeper into the problem of how bacteria divide. Of course, none of this would have been possible without the brilliance of Diamond’s beamlines and their staff, to whom we are ever grateful.”
Research into the atomic structures of biological organisms is becoming ever more achievable as scientific technology advances. Powerful scientific machines like Diamond allow scientists to see things that are far too small to be glimpsed with a standard microscope. Over the course of its lifetime, Diamond has supported the solving of over 2000 protein structures such as this.
Uncovering this particular piece in the puzzle could have significant implications for future antibiotic targets, and the discovery of the EzrA structure contributes to our understanding of a vital field: the significance of miniscule proteins in the workings of our bodies. These intricate and elegant structures are responsible for critical biological processes, and findings such as this are vital to our wider understanding of disease and healing.
Related topics
Antibiotics