Mouse study demonstrates that gut microbes protect against sepsis
Posted: 23 February 2018 | Drug Target Review | No comments yet
A recent study at the University of Pennsylvania has identified that introducing particular microflora to the blood of mice increases immunoglobin A (IgA) levels, which appears to aid survival against sepsis.
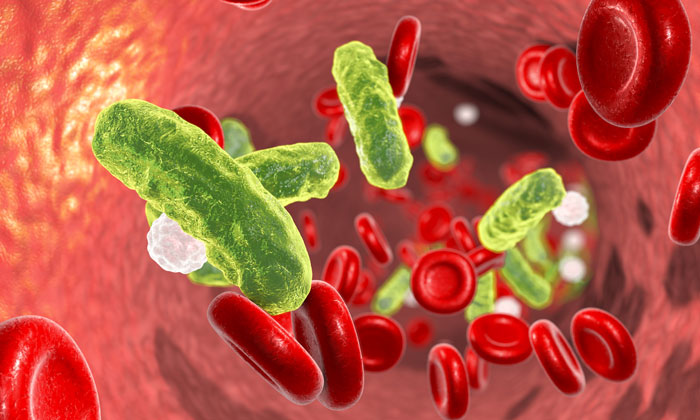
Sepsis occurs when the body’s response to the spread of bacteria or toxins to the bloodstream damages tissues and organs. New research indicates that the fight against this disease could include the help of gut bacteria. Penn scientists have found that giving mice particular microbes increased blood levels of immunoglobulin A (IgA) antibodies, thus protecting against the kind of widespread bacterial invasion that leads to sepsis.
Prior work has made this link between gut microbes and IgA responses, and IgA specific to components of intestinal bacteria have also been detected in sera of mice. Additionally, it is known that people with IgA deficiencies are more likely to succumb to sepsis. However, whether these observations were linked in any way remained unknown.
Investigating IgA antibodies and sepsis
Previous research showed that immunoglobulin M (IgM) antibodies quickly respond to blood-borne bacteria in sepsis and that gut microbes trigger immunoglobulin G (IgG) antibody responses that can block bacterial infection. The researchers involved in this current work addressed the question of whether gut microbes could trigger IgA responses that protect against sepsis.
“We propose that serum IgA and IgG antibodies may play roles similar to the protective role proposed for natural IgM antibodies, with the IgA component providing a non-inflammatory mechanism for keeping invading bacteria in check,” says first author Joel Wilmore of the University of Pennsylvania.
To investigate this possibility, senior author David Allman of the University of Pennsylvania and his team looked at IgA antibodies, which are readily detected in mice and humans but whose role in host protection against sepsis was unknown. The researchers found that exposing mice to a unique but natural microflora that included several members of the Proteobacteria phylum led to increases in IgA levels in the blood. Additionally, they discovered that shifting the mouse gut to a Proteobacteria-rich microbiota led to IgA-mediated resistance to sepsis in mice.
When the researchers transferred blood lacking IgA into mice with sepsis, all but one animal died within two days. By contrast, mice that received blood enriched with IgA survived much longer. Taken together, the findings suggest that commensal microbes can have a substantial impact on IgA levels in the blood, resulting in protection against bacterial sepsis.
Based on these findings, the researchers plan to further dissect the mechanism by which IgA confers protection against sepsis and explore ways to harness the specific properties of these antibodies to develop a treatment that may be applied to the human form of the disease. In the meantime, they urge caution against over-interpreting the new findings.
Proceed with caution
“The study is limited by the fact that the microbiome in every person or animal is unique to some degree, and our study is in the context of the animal facility at the University of Pennsylvania,” Allman says. “While IgA protected mice in our study, it should not be assumed that IgA could replace standard treatments provided to patients in a clinical setting.”
Related topics
Antibodies, Disease Research
Related conditions
Sepsis
Related organisations
Pennsylvania University
Related people
David Allman, Joel Wilmore