NanoMIPs efficiently target and eliminate cancer cells
Posted: 24 August 2018 | Dr Zara Kassam (Drug Target Review) | No comments yet
A joint research team has demonstrated the possibility of developing a new type of anti-neoplastic drugs based on nanoMIPs…
A joint research team from Russia and the UK has demonstrated the possibility of developing a new type of anti-neoplastic drugs based on nanoMIPs, or “plastic antibodies.” NanoMIPs are synthetic polymers that can function as antibodies, selectively binding to target proteins on the surface of cancer cells. This approach could lead to a paradigm shift in the development of new methods for cancer treatment. The research was carried out by an international team from the University of Leicester, University College London, the Institute of Cytology of the Russian Academy of Sciences, and the Moscow Institute of Physics and Technology.
The main drawbacks of most anti-cancer medications are their low specificity and the associated side effects. Conventional chemotherapy targets all dividing cells without exception, thus both healthy and cancer cells are affected.
However, the advances in cancer research have led to a better understanding of the molecular mechanisms and the primary agents responsible for tumour development. New findings have enabled novel anti-neoplastic drugs that can distinguish between healthy and cancer cells by acting on specific molecular targets. Because the cells in tumours undergo rapid division, they rely on a constant supply of substances stimulating cellular growth and proliferation. These substances, known as growth factors, come from outside the cell and can be identified by corresponding receptor proteins on the cell surface. These external factors activate intracellular signalling, stimulating cancer cell proliferation. It turned out that the receptor proteins on the cell surface are often overexpressed — that is, synthesized in excess — in various solid tumours
.Over the past two decades, a series of therapeutic drugs acting on growth factors have been developed and clinically tested. The new medications suppress the binding of growth factors to the receptors, as well as directly affecting their enzyme activity. It is not surprising that the development of new synthetic drugs against this type of targets is a promising area of molecular pharmacology attracting close attention of researchers all over the world.
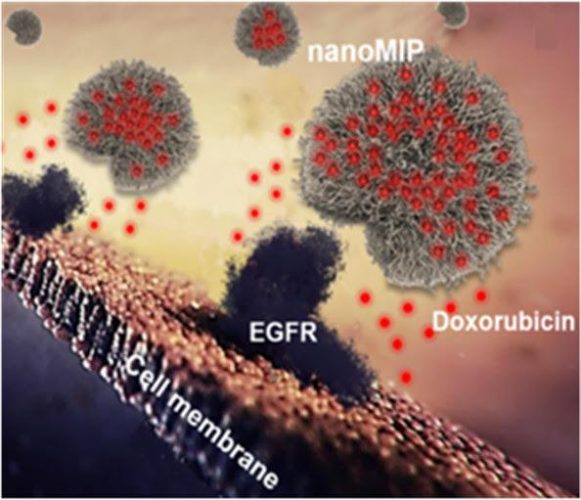
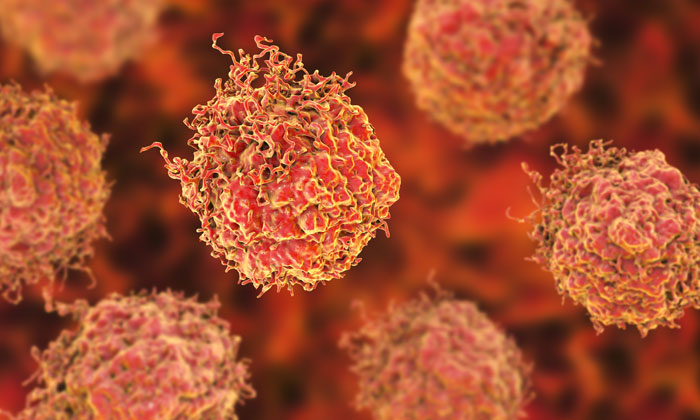
Synthetic polymer nanoparticles, or nanoMIPs, bind to cell surface via the epidermal growth factor receptor (EGFR). The red dots represent the cytotoxic agent doxorubicin, which is delivered by nanoMIPs and penetrates the cell membrane. (F. Canfarotto et al./Nano Letters)
The international research group led by Professor Nickolai Barlev, the head of the Laboratory of Cell Signaling Regulation at MIPT, has shown that it is possible to develop a new class of anti-neoplastic medicines based on a kind of particles called nanoMIPs, or nanosized molecularly imprinted polymers. NanoMIPs are a synthetic polymer alternative to antibodies with a 3D structure that enables them to bind only to a certain fragment of a target protein. This ensures their high specificity. Unlike antibodies, nanoMIPs can also carry additional anti-cancer agents. In their research, the authors proved for the first time that it is possible to synthesize nanoMIPs capable of selectively binding to the amino acid sequences of their target proteins. The study has also demonstrated the potential for nanoMIP application in targeted drug delivery.
NanoMIPs are synthesized in the presence of a target protein, which leaves a “mark” on the nanoparticle. This process is called imprinting and it can be compared to mould casting — the end product takes on the shape of the original template. Through this process, nanoMIPs acquire the ability to selectively recognise the target molecule and bind to it.
The target used by the authors of the study is the epidermal growth factor receptor (EGFR). This protein is overexpressed in many types of tumours
associated with colorectal, lung, brain, and breast cancer, including its most aggressive form, the triple-negative breast cancer. For this reason, EGFR served as one of the first targets for antibody-based anti-neoplastic medicines.
The team worked with nanoparticles obtained using a double-imprinting approach against two target molecules: a cytotoxic drug called doxorubicin and a linear epitope of EGFR. (An epitope is the part of a target molecule that is recognised by the antibody which binds to it.) Therefore, the final product both binds EGFR and delivers therapeutics to cancer cells.
“Although they are efficient in clinical use, antibody-based drugs are difficult to design and expensive to produce.
Tumours with EGFR overexpression are successfully treated with specific monoclonal antibodies targeting this receptor (cetuximab, or Erbitux®). However, because the drug is unstable, new doses of antibodies have to be administered for the entire period of treatment. And the total cost of a course of therapy can be as high as $100,000. Synthetic antibody alternatives, such as nanoMIPs, do not have these limitations. Moreover, unlike biomolecules, their stability does not depend on temperature and acidity, which means they have a much wider range of potential applications. Looking forward, they could expand the range of options available for diagnostics and treatment of many diseases,” says Prof Barlev, who is the senior author of the study.
What is more, the synthesis of selective nanoMIPs does not necessarily require the imprinting of the whole cell. Rather, only a specific part needs to be imprinted. This small part — a short oligopeptide — is attached to glass beads via covalent chemical bonds. The beads are then mixed with acrylamide monomers and doxorubicin. It should be noted that polyacrylamide, unlike its monomers, is biologically harmless and is used, for example, to produce soft contact lenses. When the temperature is increased, the monomers begin to polymerise, forming particles that are 100-200 nanometers large, incorporate doxorubicin, and carry a molecular imprint of the target protein. Unreacted monomers and nonspecific nanoparticles are eluted, while the synthesised “plastic antibodies” remain bound to the glass beads.
“For the first time, we have produced polyfunctional nanoMIPs capable of selective target protein recognition and suitable for specific drug delivery. This used to be impossible, because the available technology for nanoMIP synthesis did not allow us to standardise the conditions in which the particles were obtained, so the efficiency of the end product was unpredictable. We solved this problem by using solid-phase synthesis. Our next goal is to create ferromagnetic nanoMIPs, which would considerably expand the diagnostic and therapeutic potential of our ‘plastic antibodies,'” Prof Barlev comments.
The results of the study have also revealed moderate and specific toxicity of nanoparticles against tumour
cells. Notably, the toxicity was entirely due to doxorubicin incorporation during the polymerisation process, as the control nanoparticles, which did not contain the anti-cancer drug, did not have any effect on the cells. In addition, when therapeutic nanoMIPs were administered, the cells developed multiple DNA breaks, which are a characteristic reaction to the effect of doxorubicin. Finally, the binding of the “plastic antibodies” to EGFR led to a decrease in the density of receptors on the cell surface.
The potential therapeutic effects of nanoMIPs for treating EGFR-dependent tumours is ultimately attributable to three factors: the direct cytotoxic effect of the anti-neoplastic drug delivered to the cell, the masking of the receptor from the ligand, and the reduction of EGFR concentration on the cell surface. Successful in vitro experiments suggest that nanoMIPs hold promise as vehicles for targeted drug delivery and call for further research.
The paper reporting the results of the study was published in Nano Letters.
Related topics
Antibiotics, Antibodies, Disease Research, Immunology, Oncology, Protein
Related conditions
Cancer
Related organisations
Leicester University, Moscow Institute of Physics and Technology, the Institute of Cytology of the Russian Academy of Sciences, University College London
Related people
Professor Nickolai Barlev