In-depth research maps virulence factor of the influenza A virus in real time
Posted: 21 May 2019 | Drug Target Review | No comments yet
Findings from a recent study could lead to the development of therapeutic approaches against influenza A viruses.

Researchers at Kanazawa University have studied the structure of one of the virulence factors – haemagglutinin (HA) – of avian influenza virus, H5N1. They did this using high-speed atomic force microscopy (HS-AFM) and the findings are essential for developing therapeutic approaches against influenza A viruses in the future.
Understanding the structure and properties of HA when it is initially synthesised by host cells in its precursor form (HA0) is paramount to deciphering HA. So Dr Richard Wong, senior author of the study, and his research team visually analysed the recombinant HA0 protein of H5N1 with the HS-AFM system developed by Kanazawa University.
Both HA0 and HA exist in homotrimeric forms and conversion of HA0 to HA does not significantly modify the homotrimeric structure. Therefore, it is sensible to use HA as a template to generate HA0 HS-AFM simulation images. Acidic endosomal environment is the critical factor for HA to induce fusion between the viral membrane and endosomal membrane in order to release viral materials into host cells.
To elucidate the acidic effect on HA0, it was first exposed to an acidic environment. The trimer of HA0 turned out to be very sensitive to the acidic solution and expanded considerably. When conformational changes of haemagglutinin were measured in real-time using HS-AFM, the team found that its area was larger, and its height shorter. Acidic environment essentially made the molecule flatter and more circular, as compared to its original counterpart. This change in conformation was, however, reversible as the structure reverted back to its original form upon neutralisation.
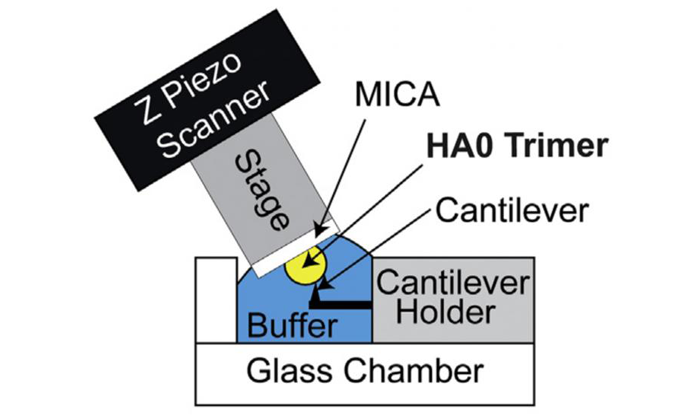
HS-AFM setup for direct visualisation of HA0 trimer. Schematic diagram of the HS-AFM setup for scanning the HA0 trimer (credit: Kanazawa University).
“Our pilot work establishes HS-AFM as an inimitable tool to directly study viral protein dynamics, which are difficult to capture with low signal-to-noise techniques relying on ensemble averaging, such as cyro-EM and X-ray crystallography,” said lead author of the study Dr Kee Siang Lim. “With high scanning speed and a minimally invasive cantilever, we predict that HS-AFM is feasible to reveal the flow of irreversible conformational changes of HA2 induced by low pH, which is mimicking the true biological events that occur when HA enters a host endosome, in future study.”
This study paved the way for investigating biological events within viruses in real-time. “Our work establishes HS-AFM as an inimitable tool to directly study viral protein dynamics, which are difficult to capture with low signal-to-noise techniques relying on ensemble averaging, such as cyro-EM and X-ray crystallography,” added Dr Richard Wong.
The research can be found on Science Direct.
Related topics
Crystallography, Imaging, Protein
Related conditions
Influenza
Related organisations
Kanazawa University
Related people
Dr Kee Siang Lim, Dr Richard Wong