Targeting a blood stem cell subset shows lasting gene editing
Posted: 31 July 2019 | Drug Target Review | No comments yet
Scientists report how editing a portion of stem cells with CRISPR-Cas9 is sufficient for long-term reactivation of therapeutic haemoglobin.
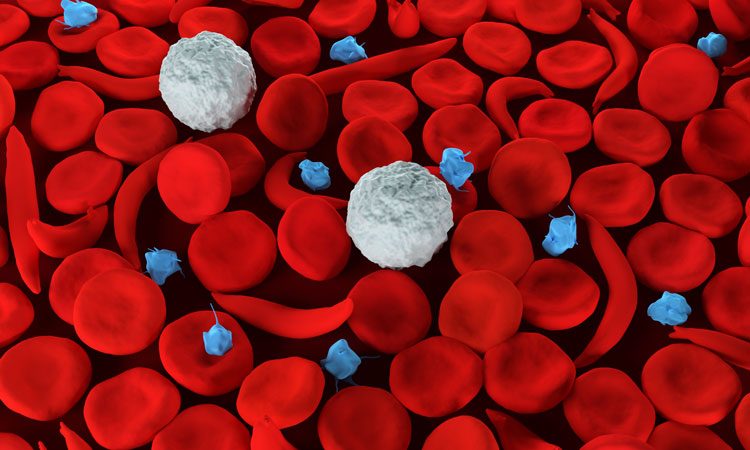
Researchers have used CRISPR-Cas9 for long-lived blood stem cell editing to reverse the clinical symptoms observed with several blood disorders, including sickle cell disease and beta-thalassemia.
The proof-of-principle study suggests that efficient modification of targeted stem cells could reduce the costs of gene-editing treatments for blood disorders and other diseases while decreasing the risks of unwanted effects that can occur with a less discriminating approach.
“By demonstrating how this select group of cells can be efficiently edited for one type of disease, we hope to use the same approach for conditions such as HIV and some cancers,” said senior author Dr Hans-Peter Kiem, director of the Stem Cell and Gene Therapy Program and a member of the Clinical Research Division at Fred Hutchinson Cancer Research Center.
For this pre-clinical study (which is expected to lead to human trials) the researchers picked a gene related to sickle cell disease and beta-thalassemia. The researchers then used CRISPR-Cas9 gene editing to remove a piece of genetic code that normally turns off foetal haemoglobin proteins.
Snipping this control DNA with CRISPR enables red blood cells to continuously produce elevated levels of foetal haemoglobin.
The edits were taken up efficiently by the targeted cells: 78 percent took up the edits in the lab dish before they were infused. Once infused, the edited cells engrafted, multiplied and produced blood cells, 30 percent of which contained the edits. This resulted in up to 20 percent of red blood cells with foetal haemoglobin, the type of haemoglobin that reverses disease symptoms in sickle cell disease and thalassemia.
The scientists also believe that carrying out genetic fixes on the smaller pool of cells required for therapeutic benefit will lessen safety concerns and reduce the risk of off-target effects.
Snipping this control DNA with CRISPR enables red blood cells to continuously produce elevated levels of foetal haemoglobin.
This was the first study to specifically edit a small population of blood cells that Kiem’s team previously identified as being solely responsible for regrowing the entire blood and immune system, CD90 cells.
The self-renewing properties of this population of stem cells make them a potential candidate to deliver gene therapy because they can provide long-term production of these genetically modified blood cells and thus could cure a disease. Since they represent just five percent of all blood stem cells, targeting them with gene-editing machinery would require fewer supplies and potentially be less costly.
The study was published in Science Translational Medicine.
Related topics
CRISPR, Gene Therapy, Stem Cells
Related conditions
sickle cell disease, thalassemia
Related organisations
Fred Hutchinson Cancer Research Center
Related people
Dr Hans-Peter Kiem