‘Tricking’ bacteria could lead to more effective antibiotics
Posted: 11 October 2019 | Rachael Harper (Drug Target Review) | No comments yet
A technique to ‘trick’ bacteria into revealing pores in their cell walls has been developed and targeting these could make antibiotics more effective.
A new technique to ‘trick’ bacteria into revealing hundreds of holes in their cell walls has been developed. According to the researchers. this could lead to the creation of drugs that destroy bacteria’s cells.
Targeting these pores could make current antibiotics more effective or allow for the development of antibiotic-free drugs that can use these openings.
When subjected to certain stimuli pores in the cell membranes open up to allow liquid to flood out of the cell to prevent it from bursting. These act as a gateway to deliver treatments that destroy the bacteria’s cells. The largest of these gated-pores is known as the Mechanosensitive Channel of Large conductance (MscL).
In this latest study, researchers have found a way to trick bacteria’s cell walls into opening these channels, making the bacteria much more vulnerable to drugs.
“Through understanding the gateways in the bacteria’s cell walls, we can then control their opening and closure,” said Dr Christos Pliotas from the University of Leeds, UK, who led the study. “Additionally, existing antibiotics should become more efficient by facilitating their access to the cell through MscL pores resulting in increased antibiotic concentration inside the cytoplasm.”
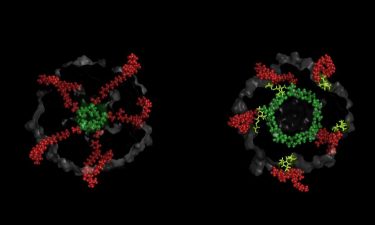
An artist rendering of opened bacterial cell wall pore (credit: University of Leeds).
This study shows for the first time that MscL channels are being kept closed by membrane lipids and demonstrates that when access of these lipids is disrupted by molecular nano-guards engineered at the entrance of the nano-pockets, the channel opens its pore.
The MscL is found in all bacterial pathogens and archaea, but absent from humans. Therefore, selective targeting of this channel would leave human cells intact.
“This study used an emerging EPR method called PELDOR (or DEER),” added co-author Dr Bela Bode, University of St Andrews. “We introduce tiny chemical markers on MscL and monitor changes in their distances. This has been instrumental for understanding the stimulus for opening of these complex biological systems.”
The study was published in Nature Communications.
Related topics
Antibiotics, Cell Cultures, Drug Targets, Research & Development, Targets
Related organisations
Leeds University, University of St Andrews
Related people
Dr Christos Pliotas