Structure of lipin enzyme responsible for triglycerides revealed for first time
Posted: 18 March 2020 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have obtained the crystal structure of the lipin enzyme, the regulator of triglyceride production, which they say could inform research into heart disease, obesity and diabetes.
The structure of a lipin enzyme, which carries out an important step in the production of triglycerides, the main reservoir for long-term energy storage, has been developed to help scientists better understand how these enzymes regulate the production of triglycerides.
Led by Dr Mike Airola, of the Department of Biochemistry and Cell Biology in the College of Arts and Sciences and Renaissance School of Medicine at Stony Brook University, US, the study provides scientists with insights as to why mutations in lipins cause a loss of activity that leads to abnormal production of triglycerides implicated in heart disease, obesity and diabetes.
Lipins complete the second-to-last step of triglyceride production. However, when mutations interrupt lipin functions, the body loses its ability to store fat properly, thus potentially triggering a wide variety of metabolic-related conditions. Now, Airola and colleagues have successfully explained in the paper the first crystal structure of a specific enzyme called lipin PAP.
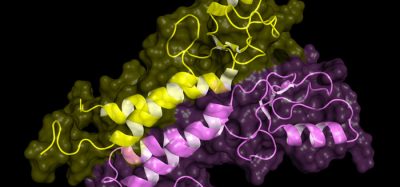
“This structure answers a long-standing question for how two essential regions, N-lip and C-lip, which are located on opposite ends of this protein in humans, come together to form a functional enzyme to help make triglycerides,” said Airola, Assistant Professor in the Department of Biochemistry & Cell Biology. “Using this structure also helps us understand how the protein interacts with membranes, which is key to regulating its activity and the production of triglycerides.”
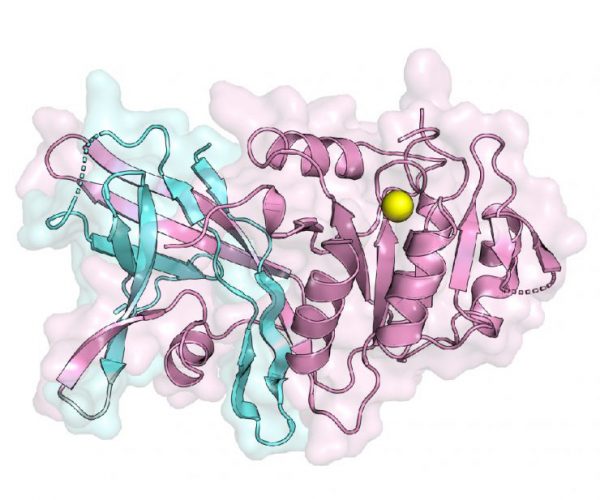
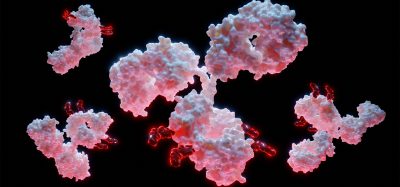
A first look at the structure of the lipin enzyme. The structure shows how two essential regions (colored blue and pink), located on opposite ends of the protein in humans, come together to form a functional enzyme to help make triglycerides [credit: Stony Brook University].
The team used X-ray crystallography, mass spectrometry and biochemistry to visualise the structure, which represents the active state of a lipin enzyme during the production of triglycerides, as well as its other functions namely lipoprotein assembly and cellular signalling.
Valerie Khayyo, a Stony Brook graduate student and first author of the study, added that the structure enables researchers to understand and see specific mutational changes in the amino acid building blocks of lipins that result in disease. She noted that mutations in lipins are increasingly being identified in patients with muscle disorders as well, such as statin-induced myopathy and extreme cases in childhood rhabdomyolysis.
Overall, Airola says that the structure and study consolidates many previous observations into a unifying framework and sets the stage for scientists to resolve several remaining important questions concerning how lipins are regulated.
The study is published in Nature Communications.
Related topics
Crystallography, Enzymes, Imaging, Lipidomics, Lipids, Mass Spectrometry, Research & Development, Structural Biology
Related conditions
Diabetes, Heart disease, Obesity
Related organisations
Stony Brook University
Related people
Dr Mike Airola, Valerie Khayyo