Hepatitis C drugs could combat SARS-CoV-2, researchers find
Posted: 17 November 2020 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have identified hepatitis C drugs that can inhibit the SARS-CoV-2 main protease, which enables the coronavirus to reproduce.
Researchers have discovered that several hepatitis C drugs can inhibit the SARS-CoV-2 main protease, a crucial protein enzyme that enables the novel coronavirus to reproduce. The study was conducted at the US Department of Energy’s Oak Ridge National Laboratory (ORNL).
According to the researchers, inhibiting this protease from functioning is vital to stopping the virus from spreading in patients with COVID-19. As the SARS-CoV-2 coronavirus spreads by expressing long chains of polyproteins that must be cut by the main protease to become functional proteins, this makes the protease an important drug target for researchers and drug developers.
“Currently, there are no inhibitors approved by the US Food and Drug Administration (FDA) that target the SARS-CoV-2 main protease,” said ORNL lead author Daniel Kneller. “What we found is that hepatitis C drugs bind to and inhibit the coronavirus protease. This is an important first step in determining whether these drugs should be considered as potential repurposing candidates to treat COVID-19.”
The team investigated several well-known drug molecules for potential repurposing efforts including leupeptin, a naturally occurring protease inhibitor and three FDA-approved hepatitis C protease inhibitors: telaprevir, narlaprevir and boceprevir. They then performed room temperature X-ray measurements to build a three-dimensional (3D) map that revealed how the atoms were arranged and where chemical bonds formed between the protease and the drug inhibitor molecules.
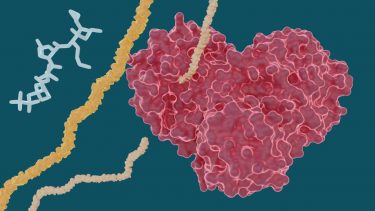
The heart-shaped SARS-CoV-2 main protease enables the virus to reproduce by cutting long chains of proteins that activate the replication process. Experiments show existing drugs used to treat hepatitis C may have potential to treat COVID-19 by stopping the “heart” of the virus [credit: Michelle Lehman, Jill Hemman/ORNL, US Dept. of Energy].
The experiments yielded promising results for certain hepatitis C drugs in their ability to bind and inhibit the SARS-CoV-2 main protease – particularly boceprevir and narlaprevir. Leupeptin exhibited a low binding affinity and was ruled out as a viable candidate.
To better understand how well or how tightly the inhibitors bind to the protease, the researchers used in vitro enzyme kinetics, which enabled them to study the protease and the inhibitor in a test tube to measure the inhibitor’s binding affinity with the protease. The higher the binding affinity, the more effective the inhibitor was at blocking the protease from functioning.
“What we’re doing is laying the molecular foundation for these potential drug repurposing inhibitors by revealing their mode of action,” said ORNL corresponding author Andrey Kovalevsky. “We show on a molecular level how they bind, where they bind and what they are doing to the enzyme shape. And, with in vitro kinetics, we also know how well they bind. Each piece of information gets us one step closer to realising how to stop the virus.”
The study also sheds light on the SARS-CoV-2 protease’s ability to change or adapt its shape according to the size and structure of the inhibitor molecule it binds to. The team found that pockets within the protease where a drug molecule would attach are highly malleable or flexible and can either open or close to an extent depending on the size of the drug molecules.
“The research suggests that hepatitis C inhibitors are worth thinking about as potential repurposing candidates. Immediately releasing our data allows the scientific community to start looking at the interactions between these inhibitors and the protease,” said ORNL corresponding author Leighton Coates.
The study is published in Structure.
Related topics
Drug Leads, Drug Repurposing, In Vitro, Lead Generation, Research & Development
Related conditions
Covid-19
Related organisations
Oak Ridge National Laboratory (ORNL)
Related people
Andrey Kovalevsky, Daniel Kneller, Leighton Coates