CRISPR tagging used to grow neuronal cells from stem cells
Posted: 3 December 2020 | Victoria Rees (Drug Target Review) | No comments yet
A new technique using CRISPR has been developed by researchers to identify programmed stem cells that mature into neuronal cells.

A team of biomedical engineers has created a new way to turn stem cells into a desired cell type using CRISPR tagging and gene regulatory networks. The study was conducted at Duke University, US.
According to the researchers, several methods already exist to programme stem cells into other cells, but often, these stem cells do not mature correctly when cultured in the lab.
“The cells might seem right at first glance, but they are often missing some of the key properties you want in those cells,” said Josh Black, who led the work in Charles Gersbach’s lab.
Using CRISPR gene editing, the researchers created a method to identify which transcription factors – master controllers of gene activity – were essential to making a good neuron. They demonstrated the potential of the approach to make mature adult neurons, but say it could be applied to programme any cell type.
When Black joined Gersbach’s lab, he investigated how to use these tools to turn on genes that could convert one cell type into another to create better disease models. In 2016, Black and Gersbach reported an approach to use CRISPR-based gene activators to turn on gene networks that would convert fibroblasts to neuronal cells. This study targeted gene networks that were known to be associated with neuronal specification, but did not generate cells with all of the properties needed to make effective disease models. However, the right gene networks to generate those desired cells were unknown and there are were thousands of possibilities encoded in the human genome. This led Black and Gersbach to devise a strategy to test all of the networks in a single experiment.
Starting with pluripotent stem cells, the team engineered some that fluoresced red once they became neuronal. The brighter the fluorescence, the stronger the push towards a neuronal fate. Then they made a pooled library of thousands of guide RNAs targeted to all of the genes that encode transcription factors in the human genome.
They introduced the CRISPR gene activator and guide RNA library into the stem cells so that each cell only received a single guide RNA and therefore turned on its particular corresponding transcription factor gene target. Then they sorted the cells based on how red they became and sequenced the guide RNAs in the most and least red cells, which told them which genes, when turned on, made the cells more or less neuronal.
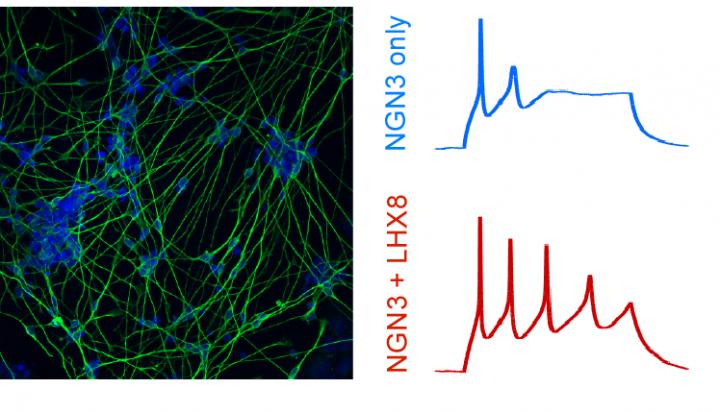
CRISPR-based activation of gene networks implicated in human stem cells becoming neuronal cells led to the generation of cells with neuronal shapes and markers (left) and enhanced function and electrophysiological properties, including producing more action potentials more frequently (right) [credit: Gersbach Lab, Duke University].
When they profiled the gene expression from the stem cells engineered with the guide RNAs, the results suggested that the corresponding cells generated more specific and mature types of neurons. They also found genes that worked together when targeted simultaneously. Moreover, the experiment revealed factors that antagonised the neuronal commitment of the stem cells and when they used CRISPR-based repressors of those genes, they could also enhance the neuronal specification.
To know if these engineered cells recapitulated the function of more mature neurons, they turned to Professor Scott Soderling. Shataakshi Dube, a grad student in Soderling’s lab, used a technique known as patch clamp electrophysiology to measure the electrical signals inside the newly formed neurons. By poking a tiny hole in the cell with a very small pipette, she looked inside the neuron and saw whether it was transmitting electrical signals known as action potentials. They found that the neurons engineered to activate a particular pair of transcription factor genes were more functionally mature, emitting more action potentials more frequently.
“I was curious but sceptical on how neuronal these stem cells could become,” Dube said, “but it was remarkable to see how much these programmed cells looked just like normal neurons.”
The process from stem cell to mature neuronal cell took seven days, dramatically shortening the timeframe compared to other methods that take weeks or months. This faster timeline has the potential to accelerate the development and testing of new therapies for neurological disorders.
The researchers say the same method for screening transcription factor genes and gene networks could be used to improve methods to make any cell type, which could be transformative for regenerative medicine and cell therapy.
“The key to this work is developing methods to use the power and scalability of CRISPR-based DNA targeting to programme any function into any cell type,” Gersbach said. “By leveraging the gene networks already encoded in our genome, our control over cell biology is dramatically improved.”
The study was conducted at Cell Reports.
Related topics
CRISPR, Induced Pluripotent Stem Cells (iPSCs), Neurons, Stem Cells
Related organisations
Duke University
Related people
Charles Gersbach, Josh Black, Professor Scott Soderling, Shataakshi Dube