New liver-on-a-chip model successfully mimics NAFLD
Posted: 11 March 2021 | Victoria Rees (Drug Target Review) | No comments yet
A liver-on-a-chip model has been developed to more fully represent the natural progression of NAFLD than previous models.
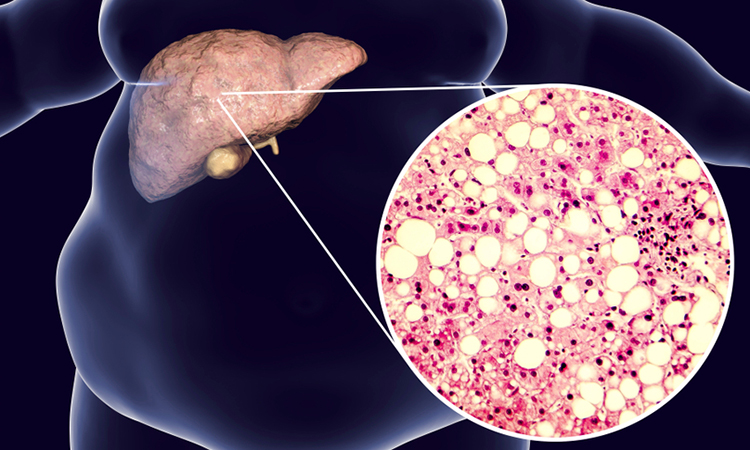
A collaborative team from the Terasaki Institute for Biomedical Innovation (TIBI), US, has developed an advanced, multicellular, structurally representative liver-on-a-chip model which mimics the full progression sequence of non-alcoholic fatty liver disease (NAFLD). The model contains all four types of human primary cells which are involved in the sequence: Kupffer cells, endothelial cells, stellate cells and hepatocytes.
According to the researchers, NAFLD is the most prevalent chronic liver disease worldwide. There is normally a small amount of fat found in the liver; however, if the amount of fat makes up five percent or more of the liver, this is considered to be NAFLD and it must be managed to avoid serious disease progression.
Abnormal accumulation of fatty acids in the liver triggers the immune cells called Kupffer cells, as well as endothelial cells lining the liver’s blood vessels, to release both signalling and chemically reactive molecules. These molecules stimulate stellate cells to synthesise and deposit fibrosis proteins, which lead to scar formation in the liver.
One of the main difficulties in developing treatments is that there are no accurate NAFLD models with which to test treatment efficacy. Animal models do not truly represent the steps involved in human NAFLD and human models developed in the past have also shown limitations. A relatively advanced “artificial fibrosis model” to stimulate the final, fibrosis-development part of the disease sequence has been created, but it did not fully correlate with observations of the full disease progression.

Liver microtissues growing as multicellular spheroids in custom-designed microwells. The spheroids are used in modelling NAFLD [credit: Khademhosseini Lab].
To address these challenges, the team mixed the four types of human primary cells together to grow multicellular microtissues in an array of pyramid-shaped microwells; the cell numbers and proportions were adjusted to yield microtissues of optimum shape to maintain nutrient and oxygen concentrations similar to naturally-occurring liver tissues. The microtissues were then enclosed within a gelatinous substance to enable the tissues to more fully match the structure and cell-to-cell interactions of native liver tissues.
The team used their multicellular liver-on-a-chip model in tests designed to validate the role of stellate cells in NAFLD. They concluded that stellate cells appear to be involved with blood vessel formation, production of chemically reactive compounds and to some degree in fatty acid accumulation. They also concluded that stellate cells may interact with Kupffer and other liver cells to produce certain signalling molecules. All these events are crucial components in full NAFLD progression.
The team also tested the effects of fatty acid addition to their model. The results indicated that the presence of fatty acids and active stellate cells accelerated inflammatory responses and increased the production of fibrotic proteins, further validating the model’s mimicry of the natural progression of NAFLD.
Subsequent tests compared the NAFLD model to the previously mentioned artificial fibrosis model driven by transforming growth factor (TGF)-β. When comparing both models, the new fibrosis model mimicked the natural progression of NAFLD by exhibiting higher levels of fat accumulation and fibrosis molecules than in the artificial fibrosis model.
In addition, the team tested the response to anti-fibrotic drugs in both models. The tests demonstrated that TIBI’s NAFLD model demonstrated a more robust drug transport and metabolic response upon delivery of the drugs.
“Our multicellular liver-on-a-chip system is a laboratory model that is more advanced and more fully represents the natural progression of NAFLD than in previous models,” said Dr Junmin Lee, one of the researchers. “This is supported by the experimental results from our comparison studies.”
“The multicellular model described here is an exemplary demonstration of the level of work that we do in developing personalised physiological models,” said Dr Ali Khademhosseini, Director and CEO of the TIBI. “It has the potential for helping to discover treatments for a prevalent and potentially fatal disease.”
The results of the study are published in Nano Micro Small.
Related topics
Disease Research, Drug Development, Organ-on-a-Chip
Related conditions
non-alcoholic fatty liver disease (NAFLD)
Related organisations
Terasaki Institute for Biomedical Innovation (TIBI)
Related people
Dr Ali Khademhosseini, Dr Junmin Lee