Ex vivo stem cell treatment a promising therapy against MRSA
Posted: 17 September 2021 | Anna Begley (Drug Target Review) | No comments yet
A new ex vivo model treated animal wounds with mesenchymal stromal cells (MSCs) and reduced MRSA infection, expanding therapeutic options for humans.
Researchers at Cornell University, US, have created an ex vivo stem cell model that can treat wounds with the secretion of a type of stem cell called mesenchymal stromal cell (MSC). The model effectively reduced methicillin-resistant Staphylococcus aureus (S aureus) – better known as MRSA – and stimulated the surrounding skin cells to build up a defence against the bacterial invader.
Currently, MRSA has become a major healthcare problem as these bacteria can become threatening under certain circumstances, such as in immunocompromised patients or in infected wound environments, and because they have grown resistant to many antibiotics.
MSCs are stem cells that can be isolated from bone marrow, adipose, blood and other tissue sources. “Initially, the use of MSCs for tissue regeneration was advocated based on their ability to differentiate into various tissue types. For this reason, it was anticipated that injected MSCs colonise the injury site, differentiate into the appropriate tissue type and regenerate the damaged tissue,” explained corresponding author Dr Gerlinde Van de Walle. “However, studies are revealing that only a small portion of administered MSCs actually incorporate into injured tissue. For this reason, it is becoming generally accepted that the beneficial effects in tissue repair and regeneration are more likely indirect, depending on the paracrine effects of what these cells secrete.”
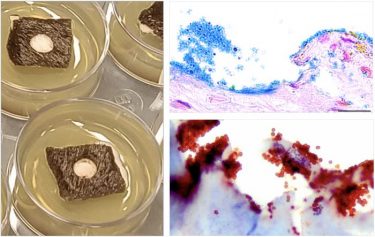
The equine ex vivo skin biofilm explant model with an image of skin explants in culture (left) and bacterial biofilms stained with Alcian Blue (upper right) and an antibacterial antibody (lower right) to show the presence of bacteria in the skin biofilm explant model. [credit: AlphaMed Press]
“This intriguing finding opens up novel therapeutic perspectives based on the development of cell-free regenerative therapies using the secretome of MSC that includes both soluble factors and factors released in extracellular vesicles,” Van de Walle continued. “Such cell-free therapies might prove safe and potentially more advantageous alternatives by overcoming the risks and obstacles associated with the allogeneic use of the cells themselves.”
The team began by establishing an equine skin biofilm model. Skin samples from horses were cultured over a period of three days from freshly harvested equine skin. They then used an MRSA-infected wound model that was created by making uniform wounds in the skin samples and inoculating them with either MRSA or its non-antibiotic-resistant counterpart, methicillin-sensitive S aureus (MSSA). The wounds were then treated for 24 hours with either antibiotics, DMEM (negative control) or MSC secretome. At the end of the treatment period, bacterial load was measured by evaluating colony-forming units (CFU) per gram of tissue.
“The results showed that secreted factors from the MSCs significantly decreased the viability of MRSA in our novel skin model,” first author Dr Charlotte Marx reported. “Moreover, we demonstrated that equine MSC secretions increase the antimicrobial activity of the skin cells by stimulating immune responses of the surrounding resident skin cells… We propose that by identifying additional effective treatments, we can contribute to reducing the use of antibiotics in both veterinary and human medicine, which is important for the fight against antibiotic resistance.”
The study was published in STEM CELLS Translational Medicine (SCTM).
Related topics
Antibiotics, Cell Cultures, Drug Targets, Microbiology, Small molecule, Stem Cells, Target Molecule, Therapeutics
Related conditions
methicillin-resistant Staphylococcus aureus (MRSA)
Related organisations
Cornell University
Related people
Charlotte Marx, Gerlinde Van de Walle