Team sheds light on the co-ordination of neural stem cell activation
Posted: 12 April 2021 | Victoria Rees (Drug Target Review) | No comments yet
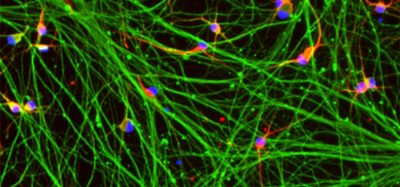
Scientists have visualised the spatial and temporal distribution of neural stem cell activation in zebrafish models.

Scientists at the Institut Pasteur and Centre national de la recherche scientifique (CNRS), both France, as well as Tel Aviv University, Israel, have successfully performed three-dimensional (3D) visualisation and spatial and temporal distribution analysis of neural stem cell activation in the adult brain of a zebrafish vertebrate model.
According to the researchers, in all adult vertebrates, neural stem cells can be recruited to produce new neurons in the brain. However, little is known about these so-called “activation” processes.
In the new study, the researchers’ findings demonstrate for the first time that activation events for these cells are co-ordinated in time and space. In particular, these results may help improve the understanding of regulation processes triggered during brain tumour formation.
Stem cells in the brain produce new neurons in adulthood. Most of the time, neural stem cells are in a dormant state known as “quiescence”. To produce neurons, they must first activate and then divide. This activation stage is crucial: it is a prerequisite for stem cell recruitment and is also critical to their survival and to the position and type of neurons formed.
Neural stem cells activate and return to a dormant state in random order and asynchronously, suggesting that these events may be co-ordinated at cell population level.
Through intravital two-photon imaging of adult fish, the scientists were able to film stem cells in their niche for several weeks and study the activation pattern of each cell in relation to neighbouring cells in real time.
The stem cells were monitored for 23 days to analyse their positions, activation events and differentiation. The fish were anesthetised and filmed using a two-photon microscope every three days.
Spatial statistical analysis and modelling performed both in real time and in long-term computational simulations demonstrated the existence of inhibitory interactions generated by activated cells, delaying the activation of other adjacent stem cells by several days. By administering a pharmacological molecule in vivo, the scientists also identified the molecular pathway involved, which is known as the Notch signalling pathway. Finally, they demonstrated that these interactions enabled steady neuronal production in time and space.
“This is the first real-time and long-term imaging of an entire neural stem cell population in the brain of an adult vertebrate. These findings demonstrate for the first time that neural stem cell activation events in the vertebrate brain are co-ordinated in time and space within the niche,” commented Laure Bally-Cuif, CNRS scientist and lead author of the study.
This study has also demonstrated that the neural stem cells themselves are involved in this co-ordination. Therefore, this research has prompted the emergence of a new concept whereby stem cell populations self-organise as a dynamic system enabling spatiotemporal co-ordination of the behaviour of each individual cell.
Such regulation may occur in tumour masses containing cancerous stem cells, in which stem cells in various states of quiescence or activation have been observed. It is also likely that such regulation takes place in the stem cell populations of other adult organs where stem cells are found in compact niches, such as epithelia.
The findings are published in Cell Stem Cell.
Related topics
Imaging, In Vivo, Neurosciences, Stem Cells
Related organisations
Centre national de la recherche scientifique (CNRS), Institut Pasteur, Tel Aviv University
Related people
Laure Bally-Cuif