Researchers have used machine learning to identify a new antimicrobial peptide that reduces inflammation, restores gut barrier function and reshapes the microbiome in ulcerative colitis.
Ulcerative colitis (UC) affects millions worldwide, causing recurrent intestinal inflammation, abdominal pain and diarrhoea. Current treatments, including 5-aminosalicylic acid, antibiotics and biologics, can control or reduce symptoms but many patients experience incomplete responses or adverse effects. Because of this, scientists are looking for safer and more effective therapies to combat it.
Now, a new study from researchers at First Hospital of Jilin University suggests that machine learning could accelerate the discovery of novel treatments, identifying antimicrobial peptides (AMPs) with potential to treat UC.
AI-driven screening discovers promising peptides
Antimicrobial peptides are naturally occurring components of innate immunity that have both antimicrobial and immunomodulatory properties. Traditionally, discovering new AMPs requires labour-intensive screening and experimental testing. In the study, Miao and colleagues developed a machine-learning pipeline combining peptide prediction models with genetic algorithms to analyse structural and physicochemical properties of over 6,000 potential candidates. The study highlighted 22 sequences that looked promising.
Five peptides were synthesised for laboratory testing. Among them, a peptide named LR, after its N- and C-terminal residues, showed the most favourable balance between antibacterial activity and low cytotoxicity. In vitro experiments demonstrated that LR had strong bactericidal effects against pathogenic bacteria including Escherichia coli and Staphylococcus aureus, while maintaining good biocompatibility, showing minimal toxicity and low haemolytic activity compared with other candidates.
Lead peptide reduces colitis in mice
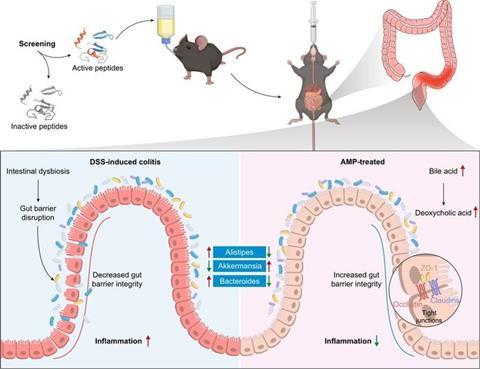
To test its therapeutic potential, LR was administered to mice with dextran sulphate sodium (DSS)-induced colitis. Treatment led to substantial improvements in disease severity, including reduced body weight loss, improved disease activity index (DAI) and less colon shortening. Histological analysis showed reduced mucosal damage and decreased infiltration of inflammatory cells. Primarily, LR treatment produced stronger effects than both the standard anti-inflammatory drug 5-aminosalicylic acid and the antibiotic ciprofloxacin in this model.
Reducing inflammation and repairing the gut barrier
Further analyses revealed that LR suppressed inflammatory responses. Levels of pro-inflammatory cytokines such as tumour necrosis factor-α (TNF-α) and interleukin-6 (IL-6) were markedly reduced after treatment. Simultaneously, the peptide helped restore intestinal barrier integrity. The expression of tight junction proteins ZO-1, claudin-1 and occludin was significantly increased, suggesting improved epithelial barrier function. These results show that LR may exert therapeutic effects both by reducing inflammation and strengthening the intestinal mucosal barrier.
Shaping the gut microbiome drives therapeutic effects
The researchers also examined LR’s impact on gut microbial communities. Sequencing of faecal microbiota revealed that treatment reshaped microbial composition in mice with colitis. The abundance of the beneficial bacterium Akkermansia muciniphila, a species linked to improved gut barrier function and reduced inflammation, increased significantly. Supplementation with A. muciniphila alone partially alleviated colitis symptoms, suggesting that microbiota modulation contributes to LR’s therapeutic effect. LR selectively inhibited pathogenic bacteria while sparing A. muciniphila, highlighting a microbiome-friendly antimicrobial profile.
Possible future therapies
The study demonstrates how integrating computational screening with experimental validation can identify stable and selective AMPs with anti-inflammatory activity in UC. While more research is needed to assess long-term safety and applicability to humans, the findings highlight a new strategy for developing microbiota-friendly therapeutics for inflammatory bowel disease.













No comments yet