A novel synthetic pathway using azide-to-diazo conversion offers pharmaceutical researchers a safer alternative to traditional diazo compound preparation. The method, eliminates hazardous reagents whilst enabling flexible access to functionalised intermediates relevant to drug candidate synthesis.

A research team from Tokyo University of Science has announced a safer and more practical approach to generating diazo compounds. These compounds are central to modern drug discovery due to their versatility in building complex organonitrogen molecules. However, their traditional preparation often depends on hazardous reagents such as diazomethane, a toxic and difficult-to-handle substance that presents some serious safety challenges, particularly in larger-scale synthesis.
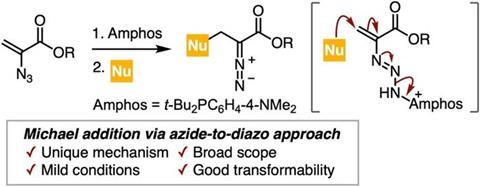
Led by Professor Suguru Yoshida, the researchers developed a method to synthesise β-heteroatom-substituted 2-diazopropionic acid esters without relying on dangerous precursors. Instead, the process uses a Michael addition, a reaction whereby one molecule donates electrons and ’attacks’ another molecule that is lacking electrons, forming a new carbon–carbon or carbon–heteroatom bond. This reaction is then mediated by an azide-to-diazo conversion, allowing access to diazo compounds from readily available starting materials under mild conditions.
“We discovered a novel form of transformation from 2-azidoacrylate esters to diazo compounds via the formation of a phosphazide intermediate and subsequent Michael addition,” says Professor Yoshida.
The innovation lies in adapting a classic reaction. The Michael addition typically involves a nucleophile adding to an electron-deficient alkene. In this case, the researchers used the reaction to convert azide groups into diazo groups through a reactive phosphazide intermediate, opening a new pathway for diazo compound formation.
From unexpected result to breakthrough
The discovery came about during investigations into how azides behave when stabilised by phosphines. By pre-treating 2-azidoacrylic acid esters with a bulky phosphine known as Amphos, the team generated a stable phosphazide intermediate. When nucleophiles such as thiols were introduced, the reaction unexpectedly produced diazo compounds rather than the anticipated azide products.
The discovery came about during investigations into how azides behave when stabilised by phosphines
This occurs because the phosphazide intermediate is more reactive than the original azide. Subsequent Michael addition triggers nitrogen–nitrogen bond cleavage, yielding the desired diazo esters efficiently.
Implications for medicinal chemistry
The method enables the introduction of heteroatoms such as sulfur and nitrogen at the β-position of the diazo ester, producing highly functionalised molecules. By varying the nucleophiles, the researchers created a broad range of diazo esters that can be further transformed into important chemical motifs, including sulfones, hydrazones and heterocycles like indoles and pyrazoles.
These structures are widely found in bioactive compounds and drug candidates, making the approach particularly relevant to drug discovery pipelines. Flexible and safer access to diazo intermediates could streamline the synthesis of compound libraries and accelerate early-stage medicinal chemistry.
Safer and more scalable chemistry
The reaction proceeds under mild conditions and avoids the use of diazomethane, improving safety and simplicity. While currently demonstrated at laboratory scale, the approach shows clear promise for broader application in pharmaceutical synthesis.
The reaction proceeds under mild conditions and avoids the use of diazomethane, improving safety and simplicity
“Diazo compounds are important intermediates widely used in the synthesis of drug candidates, functional molecules and heterocyclic compounds,” said Professor Yoshida. ”Therefore, we expect that this will contribute to the development of a wide range of research fields as a fundamental technology that enables the more practical and flexible synthesis of diverse diazo compounds.”
Next steps
The team is now working to expand the method’s scope by exploring additional starting materials, including azidoacrylamides. This could lead to an even wider range of nitrogen-containing compounds, which will be hugely beneficial to drug discovery and functional material design.
















No comments yet