New macrocycle technologies are turning a once difficult drug class into a scalable engine for developing potent oral therapies.
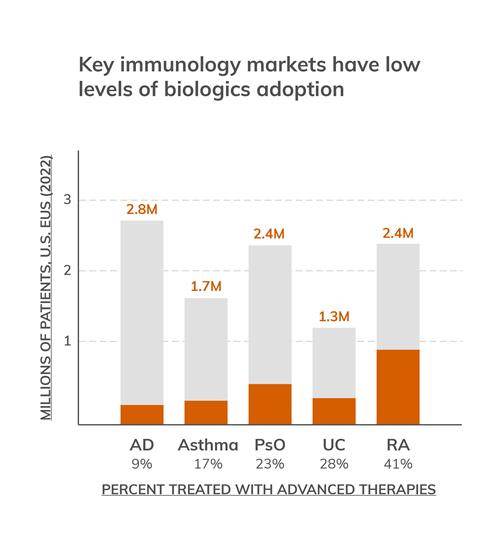
A wave of disease-causing extracellular protein targets – so far only reachable by biologics – has driven the antibody boom of recent decades, but their impact on patients has been constrained by low uptake.1 In Western countries, the high cost of goods often leads to steep drug prices and payer push back, while more broadly cold-chain logistics and complex manufacturing requirements limit their global reach. Combined with a universal strong preference for oral medications, this has resulted in only a minority of eligible patients receiving biologics treatment.

Figure 1: Key immunology markets have low levels of biologics adoption
2
[/caption]

Figure 2: Reimbursement and insurance dynamics limit biologics uptake
3
[/caption]
Now, new technologies are being developed to realise the ‘holy grail’ of the pharmaceutical industry: oral drugs that mimic the effects of biologics, often referred to as ‘oral biologics’. This new generation of therapeutics promises to leverage highly validated, effective targets in a format that is fast, cheap and simple to manufacture, store and administer.4 Oral biologics are poised to create multibillion-dollar blockbusters that redefine the standard of care for major diseases worldwide.
This fundamental shift to oral formats is driven by large pharmaceutical companies investing in technologies and strategies to increase market access through improved price positioning and patient convenience. Development of oral biologics would also help offset the looming patent cliff, with more than $200 billion in major products expected to lose exclusivity over the next five to 10 years.5,6
Macrocycles gain momentum in oral biologics
Within the broader class of oral biologics, macrocycles are an emerging hot spot of interest for big pharma. These cyclic peptides are considered a potential ‘goldilocks molecule’ with potential to combine the oral bioavailability and cell permeability of small molecules with biologics’ capability to engage challenging targets without well-defined protein pockets such as large protein–protein interactions.

Figure 3: Macrocycles occupy an ideal drug space with high oral availability and the ability to bind elusive targets
7
[/caption]
The momentum behind macrocycles in pharma is underscored by the resounding success of two late-stage, once-daily oral programmes: icotrokinra (J&J and Protagonist) and enlicitide decanoate (Merck Sharp & Dohme and Ra Pharma). Icotrokinra is a powerful anti-inflammatory that has succeeded in four Phase III trials in psoriasis and is also in development for ulcerative colitis. A new drug application has been submitted and a head-to-head study is now underway with the approved injectable biologic ustekinumab, which had nearly $11 billion in global sales in 2023.8
For its part, enlicitide decanoate has powerfully lowered ‘bad cholesterol’ in three Phase III trials through a mechanism (PCSK9 inhibition) that transformed cholesterol management with injectable drugs.9 High cholesterol affects more than 86 million Americans and is a leading cause of cardiovascular disease globally, fuelling a multi-billion-dollar PCSK9 inhibitor market.
These two candidates are striking examples of macrocycles’ vast commercial potential but interestingly they were both advanced by traditional peptide-based macrocycle medicinal chemistry – a high-risk, laborious and lengthy process with a low success rate. The resulting peptide-based macrocycles also generally have very low bioavailability. Despite their efficacy, the oral bioavailability of icotrokinra and enlicitide decanoate is in line with industry standards of one to two percent,10 impacting cost of goods, as well as fasting requirement due to the use of permeation enhancers.
Designing more orally bioavailable macrocycles
The next generation of macrocycles developed by companies such as Orbis Medicines, Unnatural Medicines, Vilya, Curve Therapeutics and Circle Pharma, among others, improve upon earlier molecules by incorporating greater chemical diversity into a smaller, highly manufacturable molecule with a better chance at good drug-like properties. By expanding beyond the 20 natural amino acids to choose from more than 6,000 synthetic ones, researchers can access a nearly infinite design space that contains more potential molecules than there are stars in the universe.11
 Figure 4: Next-generation macrocycles enable greater structural diversity within a more drug-like molecule
Figure 4: Next-generation macrocycles enable greater structural diversity within a more drug-like molecule
[/caption]
Strategic exploration of this chemical space during hit-to-lead and lead optimisation is only now becoming possible through the integration of automated, high-throughput macrocycle synthesis and screening with AI-driven computational filtering. Advances in two key technological areas, described below, have evolved macrocycle development from a high-risk, low-throughput artisanal process into a programmable, data-driven discipline.12
Solid-phase synthesis
Advances in solid-phase peptide synthesis (SPPS) have transformed macrocycle drug discovery by enabling automated, high-throughput production and screening of diverse molecular libraries.13
In traditional SPPS, each macrocycle is individually synthesised, purified and characterised – an inherently slow and resource-intensive process that limits scalability. Recent improvements in biotech discovery platforms, however, enable the synthesis of more than 1,536 peptides simultaneously in plate-based formats. Combining this with additional robust chemical modifications using solution-based chemistry allows access to tens of thousands of diverse, screening-ready macrocycles in just a few weeks’ time.14
Split-and-pool solid-phase synthesis has compounded this efficiency.15 By physically dividing resin beads into multiple reaction vessels, modifying them with different building blocks and recombining them, chemists can generate combinatorial libraries containing up to millions of unique macrocycles in under a week.
Historically, this approach has been limited by difficulties with deconvolution or the process of determining the exact structure of each macrocycle that appears as a hit in a screen. Recent advances in affinity selection–mass spectrometry now make it possible to isolate bead- or pool-derived binders, sequence or fragment them directly, and map their masses back to their corresponding building-block combinations.16 This rapid and accurate deconvolution of hits allows the full power of large combinatorial libraries to be realised.
Across the biotech industry, recent advances have focused on scaling and integrating solid-phase synthesis directly into end-to-end drug-discovery workflows.17 Automated synthesisers and robotic platforms now link synthesis and screening in a continuous discovery pipeline, evaluating binding affinity, cell permeability, proteolytic stability and other properties in parallel.
As a proof of concept, Christian Heinis at the Swiss Federal Institute of Technology Lausanne (EPFL) demonstrated an automated macrocycle design approach to engineer a thrombin inhibitor with 18 percent oral bioavailability in rats – a marked improvement over the industry standard of ~1-2 Percent.18 Published in Nature Chemical Biology, this research underpins Orbis Medicines’ discovery platform and highlights the potential of next generation discovery platforms to deliver orally bioavailable macrocycles against virtually any protein target.
AI-guided drug discovery
As biotech companies generate increasingly large and complex datasets, artificial intelligence (AI) and machine learning (ML) are becoming essential tools to accelerate and refine drug design. Generative AI models can now be customised for specific discovery goals and trained on a combination of public and proprietary data to enhance predictive accuracy and analytic power.19
Despite this progress, current generative models in the life sciences largely excel at predicting molecular binding rather than functional activity. Even binding predictions remain challenging for macrocycles and peptides containing synthetic amino acids, as structural and biophysical data in these chemical spaces remain limited.20
Proprietary experimental data are therefore emerging as a critical differentiator in the performance and reliability of AI-driven drug discovery. Early studies of large generative models indicate that training predominantly on AI-generated data can degrade model quality over time,21 underscoring the need for experimentally validated, real-world data.
In the macrocycle field, this means that access to expansive, high-quality datasets derived from automated solid-phase synthesis and high-throughput screening will be essential to building next-generation predictive models.
Companies working in this space are now using real-world data to train AI systems that guide:
- hit identification – using target structural data (eg, X-ray crystallography) as well as computational protein structure prediction to generate candidate macrocycles predicted to bind targets with high affinity.
- hit-to-lead optimisation – designing new analogues that improve upon initial hits across multiple parameters, including cell permeability and oral bioavailability. Because macrocycles do not obey standard drug development guidelines that have successfully been applied to small molecules, predicting developability is more complex. In particular, AI models must account for macrocycles’ ability to change shape and polarity depending on their environment, which impacts cell permeability and other drug-like properties.
- structure–function prediction – building models that learn from experimental functional data to link molecular design directly to biological activity.
Together, these advances mark a new era in macrocycle drug development, where automated synthesis and screening, and AI-powered design operate in continuous feedback, transforming discovery into a self-improving, data-driven process.
The Goldilocks moment: converging innovation and opportunity in macrocycles
Next-generation macrocycles have arrived at a ‘Goldilocks moment’ – when the convergence of new technologies and market need are in alignment to support the rapid advancement of a modality long considered beyond the reach of traditional drug development.
 Figure 5: Integration of physical and virtual technologies enables more effective macrocycle design
Figure 5: Integration of physical and virtual technologies enables more effective macrocycle design
[/caption]
The near-term objectives are clear:
- Demonstrate how emerging technologies can dramatically improve the efficiency and precision of macrocycle design
- Deliver macrocycles with superior oral bioavailability compared to first-generation molecules
- Translate the inherent advantages of macrocycles to accelerate the industry’s broader shift from injectable biologics to oral therapies, ultimately expanding access and convenience for patients worldwide
- Through oral macrocycles with higher oral bioavailability, enable the development of fixed dose combinations of two drugs thereby creating oral “bispecific” drugs which can drive further efficacy in hard-to-treat indications – while preserving lower pricing, patient access and global distribution to patients worldwide.
The once-unimaginable vastness of macrocycle chemical space is becoming tractable. Innovators are combining automated systems capable of large-scale synthesis, screening and analysis, with AI-driven platforms that enable predictive modelling across unprecedented chemical diversity. What was once an aspirational frontier is now a tangible opportunity to realise the full therapeutic potential of macrocycles.
About the author
 Morten Graugaard is the CEO of Orbis Medicines, a biotechnology company transforming potent biologic therapies into more accessible, orally delivered macrocycle medicines. He brings more than 20 years of leadership experience across venture investing, R&D, business development, M&A and company building.
Morten Graugaard is the CEO of Orbis Medicines, a biotechnology company transforming potent biologic therapies into more accessible, orally delivered macrocycle medicines. He brings more than 20 years of leadership experience across venture investing, R&D, business development, M&A and company building.
Before joining Orbis full-time, Morten served as its board chair and was previously a partner at Novo Holdings, where he led Novo Seeds and helped launch and scale multiple high-impact biotech companies, including Orbis Medicines.
Under his leadership, Orbis has raised more than €116 million to advance its AI-enabled discovery platform and pipeline of next-generation oral macrocycles designed to replace injectable biologics. Morten holds an MSc in human biology from the University of Copenhagen and has served on the boards of several biotechs including Muna Therapeutics, NMD Pharma, Syndesi Therapeutics and Acesion Pharma.
References:
- Makurvet FD. (2020). Biologics versus small molecules: Drug costs and patient access. Medicine in Drug Discovery, 9(9), 100075. https://doi.org/10.1016/j.medidd.2020.100075
- Sanofi. (2023). R&D Day 2023 investor presentation [PDF presentation]. https://www.sanofi.com/assets/dotcom/content-app/events/investor-presentation/2023/r-and-d-day-2023/Presentation.pdf
- Loiselle AR, Chovatiya R, Thibau IJ, et al. (2024). Evaluating Access to Prescription Medications in the Atopic Dermatitis Patient Population in the USA. Dermatology and Therapy. https://doi.org/10.1007/s13555-024-01205-0
- Zeng H, Ma M, Peng X, et al. (2025). Oral Delivery Strategies for Biological Drugs. Small Methods, 9(7). https://doi.org/10.1002/smtd.202401355
- Next drug patent cliff to challenge Big Pharma strategy. (2025, July 5). European Pharmaceutical Review. https://www.europeanpharmaceuticalreview.com/news/261834/next-drug-patent-cliff-to-challenge-big-pharma-strategy/
- Möller C, Kristic S, Walker G-C, et al. (2025, February 18). Biopharma’s Patent Cliff Puts Costs Front and Center. BCG Global. https://www.bcg.com/publications/2025/patent-cliff-threatens-biopharmaceutical-revenue?
- Peterson ML. (2017, October 15). The Evolution of Macrocycles in Drug Discovery: From Technologies to Drugs. Americanpharmaceuticalreview.com. https://www.americanpharmaceuticalreview.com/Featured-Articles/343609-The-Evolution-of-Macrocycles-in-Drug-Discovery-From-Technologies-to-Drugs/
- Icotrokinra results show potential to set a new standard of treatment in plaque psoriasis. (2025, March 8). JNJ.com. https://www.jnj.com/media-center/press-releases/icotrokinra-results-show-potential-to-set-a-new-standard-of-treatment-in-plaque-psoriasis
- Merck’s Investigational Oral PCSK9 Inhibitor Enlicitide Decanoate Met All Primary and Key Secondary Endpoints in Adults with Hypercholesterolemia in Pivotal CORALreef Lipids Study - Merck.com. (2025, September 30). Merck.com. https://www.merck.com/news/mercks-investigational-oral-pcsk9-inhibitor-enlicitide-decanoate-met-all-primary-and-key-secondary-endpoints-in-adults-with-hypercholesterolemia-in-pivotal-coralreef-lipids-study/
- Nicze M, Borówka M, Dec A, et al. (2024). The Current and Promising Oral Delivery Methods for Protein- and Peptide-Based Drugs. International Journal of Molecular Sciences, 25(2), 815–815. https://doi.org/10.3390/ijms25020815
- Morten Graugaard – CEO, Orbis Medicines. (2025, September 15). PharmaBoardroom. https://pharmaboardroom.com/interviews/morten-graugaard-ceo-orbis-medicines/
- Garcia Jimenez D, Poongavanam V, Kihlberg J. (2023). Macrocycles in Drug Discovery─Learning from the Past for the Future. Journal of Medicinal Chemistry, 66(8), 5377–5396. https://doi.org/10.1021/acs.jmedchem.3c00134
- Terrett NK. (2010). Methods for the synthesis of macrocycle libraries for drug discovery. Drug Discovery Today: Technologies, 7(2), e97–e104. https://doi.org/10.1016/j.ddtec.2010.06.002
- Habeshian S, Merz ML, Sangouard G, et al. (2022). Synthesis and direct assay of large macrocycle diversities by combinatorial late-stage modification at picomole scale. Nature Communications, 13(1). https://doi.org/10.1038/s41467-022-31428-8
- Morimoto J, Kodadek T. (2015). Synthesis of a large library of macrocyclic peptides containing multiple and diverse N-alkylated residues. Molecular BioSystems, 11(10), 2770–2779. https://doi.org/10.1039/c5mb00308c
- Lee MA, Brown JS, Farquhar CE, et al. (2025). Affinity selection–mass spectrometry with linearizable macrocyclic peptide libraries. Science Advances, 11(12), eadr1018–eadr1018. https://doi.org/10.1126/sciadv.adr1018
- Brocklehurst CE, Altmann E, Bon C, et al. (2024). MicroCycle: An Integrated and Automated Platform to Accelerate Drug Discovery. Journal of Medicinal Chemistry, 67(3), 2118–2128. https://doi.org/10.1021/acs.jmedchem.3c02029
- Merz ML, Habeshian S, Li B, et al. (2023). De novo development of small cyclic peptides that are orally bioavailable. Nature Chemical Biology, 1–10. https://doi.org/10.1038/s41589-023-01496-y
- Gangwal A, Ansari A, Ahmad I, et al. (2024). Generative artificial intelligence in drug discovery: basic framework, recent advances, challenges, and opportunities. Frontiers in Pharmacology, 15. https://doi.org/10.3389/fphar.2024.1331062
- Hu F, Jia X, Liao W, et al. (2025). Exploring the macrocyclic chemical space for heuristic drug design with deep learning models. Communications Chemistry, 8(1). https://doi.org/10.1038/s42004-025-01686-w
- Shumailov I, Shumaylov Z, Zhao Y, et al. (2024). AI models collapse when trained on recursively generated data. Nature, 631(8022), 755–759. https://doi.org/10.1038/s41586-024-07566-y
Topics
- Artificial Intelligence (AI)
- Assays
- Biologics
- Biotherapeutics
- Companies
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Targets
- High-Throughput Screening (HTS)
- Hit-to-Lead
- Informatics
- Medicinal Chemistry
- Molecular Biology
- Morten Graugaard (CEO of Orbis Medicines)
- Neurological disorders
- Orbis Medicines
- Peptide Therapeutics
- Translational Science
















