Scientists at the University of Exeter have discovered a genetic process in the deadly hospital fungus Candida auris, which could help to develop new treatments.
Researchers at the University of Exeter have located a genetic process that could lead to the formulation of new treatments for a deadly fungal infection, Candida auris, which has forced the closure of hospital intensive care units around the world.
Candida auris is particularly dangerous for critically ill patients. While it appears to live harmlessly on the skin of an increasing number of people, those on ventilators face a high risk of infection. Once contracted, the disease carries a death rate of approximately 45 percent and can resist all major classes of antifungal drugs, making it extremely difficult to treat and eradicate from hospital wards.
A global health threat
First detected in 2008, the origins of Candida auris remain unknown. Since its emergence, more than 40 countries – including the UK – have reported outbreaks. Also known as Candidozyma auris, it has been designated a global health threat and is listed by the World Health Organization as a critical priority fungal pathogen. Cases in the UK have steadily increased in recent years.
First detected in 2008, the origins of Candida auris remain unknown.
Now, for the first time, Exeter researchers have explored how Candida auris activates its genes during infection, using a novel approach involving fish larvae. The findings offer early promise for identifying potential drug targets, or even repurposing existing medications, if the same genetic processes are confirmed in humans.
Investigating the fungus in a new way
“Since it emerged, Candida auris has wreaked havoc where it takes hold in hospital intensive care units. It can be deadly for vulnerable patients, and health trusts have spent millions on the difficult job of eradication,” said research co-leader and clinical lecturer, Hugh Gifford from the University of Exeter’s MRC Centre for Medical Mycology (CMM). “We think our research may have revealed an Achilles heel in this lethal pathogen during active infection, and we urgently need more research to explore whether we can find drugs that target and exploit this weakness.”
Studying Candida auris has been challenging due to its ability to withstand high temperatures and tolerate high salt levels. Some scientists have speculated that it could originate from tropical oceans or marine animals. To overcome these obstacles, the Exeter team developed a model using Arabian killifish, whose eggs can survive at human body temperatures.
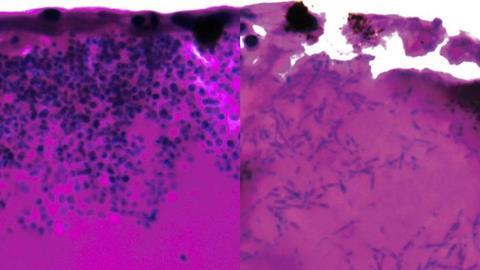
 Above image shows Candida auris filaments during infection stage. Image Credit: University of Exeter.[/caption]
Above image shows Candida auris filaments during infection stage. Image Credit: University of Exeter.[/caption] Genes reveal potential vulnerabilities
The researchers discovered that Candida auris can form elongated fungal filaments, possibly to search for nutrients. They also identified which genes are switched on and off during infection, highlighting potential vulnerabilities. Several genes activated during infection code for nutrient pumps that scavenge for iron and draw it into the fungal cells.
The researchers discovered that Candida auris can form elongated fungal filaments, possibly to search for nutrients.
“Until now, we’ve had no idea what genes are active during infection of a living host. We now need to find out if this also occurs during human infection,” said co-senior author and senior lecturer in bioinformatics at the MRC Centre for Medical Mycology, Dr Rhys Farrer. “The fact that we found genes are activated to scavenge iron gives clues to where Candida auris may originate, such as an iron-poor environment in the sea. It also gives us a potential target for new and already existing drugs.”
Innovative alternative research methods
An NC3Rs project grant supported the use of the Arabian killifish larvae model as an alternative to mice and zebrafish, offering new insights into pathogen-host interactions.
“This new publication demonstrates the utility of the replacement model to study Candida auris infection and enable unprecedented insights into cellular and molecular events in live infected hosts,” said Dr Katie Bates, NC3Rs head of research funding. “This is a brilliant example of how innovative alternative approaches can overcome key limitations of traditional animal studies.”
We now need to explore whether they could be repurposed to stop Candida auris from killing humans and closing down hospital intensive care units.
The study marks a significant step towards understanding the lethal fungus and developing strategies to combat its devastating impact in hospitals worldwide.
“While there are a number of research steps to go through yet, our finding could be an exciting prospect for future treatment. We have drugs that target iron scavenging activities,” said Dr Gifford, also an intensive care and respiratory medicine resident physician at the Royal Devon & Exeter Hospital. “We now need to explore whether they could be repurposed to stop Candida auris from killing humans and closing down hospital intensive care units.”
Topics
- Analytical Techniques
- Antifungal Therapies
- Antimicrobials
- Candida auris
- Disease Research
- Dr Katie Bates (NC3Rs head of research funding)
- Dr Rhys Farrer (senior lecturer in bioinformatics at the University of Exeter's MRC Centre for Medical Mycology)
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Repurposing
- Drug Targets
- Genetic Analysis
- Genomics & Sequencing
- Hugh Gifford (clinical lecturer at the University of Exeter’s MRC Centre for Medical Mycology)
- Infectious disease
- Microbiology
- Molecular Biology
- Neurological disorders
- the University of Exeter
- Translational Science




















