Current obesity drug development remains overly focused on short-term weight reduction, despite obesity being a chronic, multifactorial disease. Broader, mechanism-driven approaches are needed to ensure durable efficacy, safety and accessibility.
There is a somewhat narrow focus on relative weight loss when it comes to drug development for obesity. Obesity treatment requires far more than achieving short-term rapid weight loss. Obesity is a chronic disease, similar to hypertension and type 2 diabetes, but is also heterogenous and multifactorial to a greater extent than other metabolic diseases.
The development of incretin-based therapies has revolutionised obesity treatment, offering unprecedented efficacy. There are currently six approved anti-obesity medications (AOMs) which have been shown to decrease weight (compared to placebo) in the range of three to 17 percent (Table 1). Similar or greater weight loss seems to be achievable with future products that may be on the market in the next few years. However, it is important to look beyond the relative weight loss observed in clinical trials. Most patients will not have access to the same support as those in clinical trials, and as treatment will be long-term it must be affordable, safe and improve overall health.
Table 1. Approved1 medications, sorted according to overall mechanism of action.
| Medication | Placebo adjusted relative weight loss | The most prevalent side-effects (prevalence)2 |
| Targeting CNS specifically | ||
| Phentermine-topiramate (oral) | 7.8% | Paresthesia (tingling, 20%) |
| Naltrexone-bupropion (oral) | 6.4% | Nausea (33%) |
| Targeting the gut-brain axis (eg, GLP-1 and GIP receptor agonists) | ||
| Liraglutide (injectable) | 4.5% | Nausea (39%) |
| Semaglutide (injectable3) | 12.4% | Nausea (44%) |
| Tirzepatide (injectable) | 17.8% | Nausea (32%) |
| Targeting local gastrointestinal systems | ||
| Orlistat (oral) | 3% | Oily spotting (27%) Faecal urgency (22%) |
1 Approved by FDA and EMA except phentermine-topiramate, which is approved by FDA and a few European countries
2 FDA label
3 Oral semaglutide for weight management is under review by the FDA.
The main problem with obesity is not yet solved
The majority of patients that initiate medical treatment (some form of glucagon-like peptide-1 (GLP-1) receptor agonist-related therapy), discontinue treatment within the first year1 – mainly due to cost, but also side-effects and lack of treatment effect. External and internal weight stigma alongside insufficient insight regarding obesity are likely also important factors.
When obesity treatment is withdrawn, the underlying pathophysiology reasserts itself with regained weight,2,3 predominantly as increased fat mass.4 This is identical to when patients discontinue antihypertensive or lipid-lowering medications, ie, blood pressure and cholesterol levels typically return to pathological ranges. Aside from the psychological burden of multiple failed attempts to achieve sustained weight loss and considering it as an impossible mission,5 this regain has detrimental metabolic consequences beyond the simple return of weight. There is often a higher fat-to-muscle ratio than before the start of treatment6 and weight cycling (repeated loss/regain or ‘yo-yo dieting’) is associated with metabolic dysfunction, an increased risk of type 2 diabetes and cardiovascular disease.7,8
Cost – the main barrier for treatment discontinuation
Although prices for AOMs have dropped significantly, incretin mimetics such as the peptide drugs semaglutide and tirzepatide still cost around $500 per month. Generic versions of the GLP-1 receptor agonist liraglutide have started to be launched, but without any major changes in price. The high cost of treatment is partly driven by expenses related to clinical development and manufacturing and is further amplified by sustained high demand and limited supply of GLP-1 receptor agonist drugs on the market. Despite the expectation that more products would lead to lower prices, there are no clear indications that future launched products will be dramatically cheaper. Thus, the treatment cost for most current and coming AOMs will likely be beyond reach for most patients with obesity.
Side effects and lack of efficacy as important additional obstacles
Recent clinical trials using incretin-based therapies, where dose-titration is regulated and there is limited room for individualisation, have seen a substantial portion of participants experience side-effects such as nausea, vomiting and constipation – despite the intense lifestyle support provided. In clinical practice, where the possibility of more intense lifestyle support is limited, one can stretch out the dose-escalation and decrease the prevalence of at least nausea and vomiting, but still a fair number of patients cannot overcome the side effects. Furthermore, as obesity is a heterogenous disease, a sizeable minority experiences no or minimal treatment effect, even with the coming highly effective treatments or with bariatric surgery.
Long-term safety –uncertainties for most treatments
Despite incretin-based therapy (DPP4-inhibitors and GLP-1 receptor analogues) having been used for diabetes treatment for a long time, the doses employed in obesity treatment are far higher, with clinical use only dating back to late 2014 with the approval of Saxenda. Furthermore, prior to the launch of Wegovy the prescription rate of AOMs was very low. Now, as AOM use is more prevalent, side effects that were not picked up in clinical trials are emerging. Less frequent side effects will only be detected after launch, such as the increased suicide rate observed with rimonabant or the increased cancer rate seen with lorcaserin. Of the approved products, orlistat (Table 1), has the longest clinical experience as it was launched globally almost 30 years ago. Probably due to its negligible systemic uptake, the occurrence of serious adverse events is rare.
The treatment gap in obesity
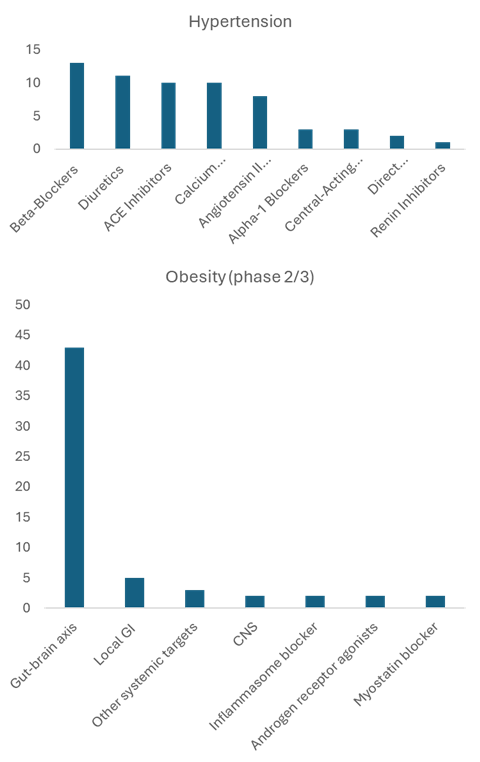
Using hypertension as an example of another chronic metabolic disease, there are currently 61 approved drugs, distributed in seven different classes of medications (Figure 1).
This affords the possibility of various combinations to maximise treatment effects and minimise side effects; it also enables more personalised treatment. The treating physician can adjust medication depending on achieved treatment effect but also side-effect profile; usually a combination of different medications is used. In contrast, there are around 56 medications in late-stage development for obesity, which are unevenly distributed in around seven different classes. More than 75 percent of projects are in the class ‘Gut-brain axis’ (Figure 1).
This skewed distribution of approved and coming medications is associated with specific problems:
- The medications within the ‘gut-brain axis’ have an overlapping side-effect profile, with a substantial portion of patients suffering from initial nausea/vomiting as well as persistent constipation. Changing to another medication within the same class will most likely not improve tolerability. Unfortunately, there is also some side-effect overlap with approved non-gut-brain medications, namely constipation and nausea (Table 1). However, combining drugs with agents that have different side-effect profiles (eg, locally acting agents or medications targeting other pathways) may be a solution.
- Given that a fair percentage of patients do not respond to gut-brain medications, changing to another medication within the same class is unlikely to improve efficacy. However, combining or replacing with agents targeting other pathways may increase treatment succuss. In addition, companion diagnostics and personalised treatment approaches are being developed to identify likely responders earlier to reduce time spent on ineffective therapies.9
- A reduction in appetite typically leads to a decrease in overall food quantity; however, for many patients, this does not necessarily translate into improved meal quality. Consequently, the risk of malnutrition and micronutrient deficiencies may increase. Locally acting enzyme inhibitors – such as the approved lipase inhibitor orlistat (marketed as Xenical) and the late-stage investigational combination EMP16 (orlistat and acarbose) – may encourage patients to select higher-quality, more nutrient-dense meals. They also have a different route of action and different side effects, enabling combination with other classes to improve efficacy and minimise side effects.
Essentially, the lack of diversity will severely limit the ability to individualise treatment, eg, by combining different medications, as it will be problematic to combine medications within the same class.

Figure 1: Hypertension and obesity, distribution of medications depending on overall mechanism of action.[/caption]
Future perspectives
As obesity is a chronic and heterogenous disease, there is a need to expand the treatment toolbox to provide different medications for different patients. The current emphasis on incretin-based therapies and novel chemical entities, while transformative, does not fully address the core challenge of obesity: the need for inclusive, sustainable and broadly accessible treatment strategies. Incretin-based drugs have demonstrated remarkable efficacy in weight reduction, yet they are not suitable or tolerable for all patients.
We suggest that a substantial ‘zone of development’ exists beyond incretin-based therapies that warrants far greater scientific and clinical investment. Expanding research into alternative mechanisms, innovative delivery systems and reformulations of established drugs will be essential to building a more resilient therapeutic landscape. Combining therapies (such as Wegovy (semaglutide), gut-brain, and EMP16 (orlistat and acarbose), local GI) – either to lower treatment costs (lower dose of expensive peptide drug), improve efficacy or mitigate adverse events – should be more widely employed. With a diversified pipeline – spanning both established and novel approaches – effective, safe and with diverse side effects, sustainable treatments can be made available to the full spectrum of individuals living with obesity.
References
- Rodriguez PJ, Zhang V, Gratzl S, et al. Discontinuation and Reinitiation of Dual-Labeled GLP-1 Receptor Agonists Among US Adults With Overweight or Obesity. JAMA Netw Open 2025;8: e2457349.
- Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab 2022;24: 1553-1564.
- Berg S, Stickle H, Rose SJ, Nemec EC. Discontinuing glucagon-like peptide-1 receptor agonists and body habitus: A systematic review and meta-analysis. Obes Rev 2025: e13929.
- Yates T, Biddle GJH, Henson J, et al. Impact of weight loss and weight gain trajectories on body composition in a population at high risk of type 2 diabetes: A prospective cohort analysis. Diabetes Obes Metab 2024;26: 1008-1015.
- Hall KD, Kahan S. Maintenance of Lost Weight and Long-Term Management of Obesity. Medical Clinics of North America 2018;102: 183-197.
- Rossi AP, Rubele S, Calugi S, Caliari C, Pedelini F, Soave F, et al. Weight Cycling as a Risk Factor for Low Muscle Mass and Strength in a Population of Males and Females with Obesity. Obesity (Silver Spring) 2019;27: 1068-1075.
- Zou H, Yin P, Liu L, Liu W, Zhang Z, Yang Y, et al. Body-Weight Fluctuation Was Associated With Increased Risk for Cardiovascular Disease, All-Cause and Cardiovascular Mortality: A Systematic Review and Meta-Analysis. Front Endocrinol (Lausanne) 2019;10: 728.
- Zou H, Yin P, Liu L, Duan W, Li P, Yang Y, et al. Association between weight cycling and risk of developing diabetes in adults: A systematic review and meta-analysis. J Diabetes Investig 2021;12: 625-632.
- Woolf EK, Diktas HE, Acosta A, et al. Precision Prevention, Diagnostics, and Treatment of Obesity: Pipedream or Reality? Obesity (Silver Spring) 2025.
Meet the authors
Ulf Holmbäck is Associate Professor in Clinical  Nutrition and Metabolism at Uppsala University. He has been in the nutrition and obesity fields as a researcher and educator for almost 30 years.
Nutrition and Metabolism at Uppsala University. He has been in the nutrition and obesity fields as a researcher and educator for almost 30 years.

Stefan Grudén, PhD (Medical Sciences, Karolinska Institutet), specialises in formulation development, innovative dosage form design and scale-up. Dr Grudén has developed product candidates from academic research to industry for Swedish life science companies and remains active in research and teaching.

Anders Forslund is a Paediatrician and Associate Professor at Women´s and Children´s health, Uppsala University. Anders has been working clinically with paediatric obesity close to 20 years and has been Head of Pediatric Obesity Unit, Uppsala University Children´s hospital since 2007. He has been active in research around obesity for almost 30 years.
Conflicts of Interest
Ulf Holmbäck, Stefan Grudén and Anders Forslund have equity interests in Empros Pharma AB, which develops the anti-obesity medication EMP16, and have acted as consultants for the company. Ulf Holmbäck, Stefan Grudén and Anders Forslund are patent holders for EMP16.
Topics
- Analytical Techniques
- Anders Forslund (Paediatrician and Associate Professor - Uppsala University)
- Cardiovascular disease
- Companies
- Disease Research
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Formulation
- Genomics & Sequencing
- Hypertension
- Informatics
- Karolinska Institutet
- Legal & Compliance
- Metabolic disorders
- Metabolomics
- Stefan Grudén (Specialist in formulation development - Karolinska Institutet)
- Translational Science
- Ulf Holmbäck (Associate Professor in Clinical Nutrition and Metabolism at Uppsala University)
- Uppsala University
- Uppsala University Children’s Hospital