Researchers at McMaster University have developed a chimeric CAR T cell therapy that successfully eliminated glioblastoma tumours in preclinical studies.
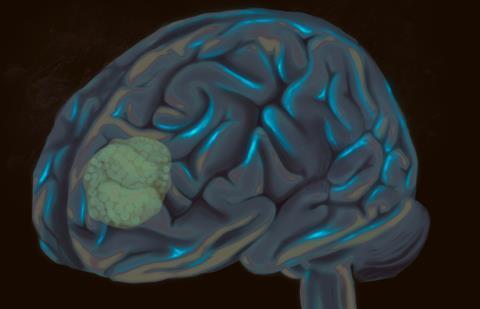
A next-generation cancer therapy developed at McMaster University has shown encouraging early results against glioblastoma, the most aggressive and most common form of primary brain cancer in adults.
In preclinical findings, researchers demonstrated that the experimental treatment was able to eliminate glioblastoma tumours that typically resist conventional therapies and rapidly return after surgery, radiation and chemotherapy.
The research could go some way to tackling a disease with a median survival time of less than 15 months after diagnosis.
“New therapies for glioblastoma are urgently needed,” says Sheila Singh, a professor in McMaster’s Department of Surgery and principal investigator on the new study. “The standard of glioblastoma care has remained largely unchanged for over two decades, and the disease remains uniformly fatal because of it.”
Targeting a key tumour protein
The experimental treatment, known as a uPAR Chimeric CAR T cell, is a form of immunotherapy designed to reprogramme the patient’s own immune system to attack cancer cells.
The therapy was developed using antibodies created in collaboration with scientists at Canada’s National Research Council in Ottawa.
Researchers focused on a protein called the urokinase receptor, or uPAR, which is found on the surface of glioblastoma cells. The protein also appears on nearby support cells that help tumours grow and survive.
Researchers focused on a protein called the urokinase receptor, or uPAR, which is found on the surface of glioblastoma cells
By targeting uPAR, the therapy attacks cancer cells directly and disrupts the biological environment that allows glioblastoma to recur after treatment.
The findings are part of a wider movement in cancer research to investigate uPAR as a therapeutic target. Singh noted that researchers at Memorial Sloan Kettering Cancer Center and Columbia University have also recently identified the protein as a promising target in lung and pancreatic cancers.
She said the growing interest in uPAR is creating opportunities for collaborative work that could eventually lead to treatments for several difficult-to-treat cancers.
Clinical trial discussions underway
Singh said discussions are already taking place about advancing the therapy towards clinical trials.
“This is a very exciting clinical candidate,” said Singh, who is also a professor of neuro-oncology and neurosurgery at King’s College London. “Our goal is to continue pushing this research forward, so that we can translate these preclinical results into first-in-human studies.”
Glioblastoma is very challenging to treat. Standard therapies can temporarily slow the disease but struggle to prevent it from returning. Researchers hope new immunotherapies such as CAR T cell treatments may eventually improve long-term outcomes for patients.
Researchers driven by patient impact
For William Maich, a postdoctoral fellow at McMaster and first author of the study, the possibility of helping patients through his research is a major source of motivation.
“It would be a dream come true for me to have some of my work help glioblastoma patients,” says Maich, who works in Singh’s lab. “Over the years, I’ve had the privilege of getting to know patients and their families through our bequeathal program at McMaster and being able to finally present them with an alternative to the current standard of care would be a great honour.”
It would be a dream come true for me to have some of my work help glioblastoma patients
McMaster University has patented the therapy and researchers are now exploring possible commercial partnerships and clinical pathways to move the treatment closer to patient testing.
While the therapy is still at an early stage of development, the study offers a potential way forward in a field where meaningful advances have been limited for decades.















No comments yet