A novel imaging-guided radiopharmaceutical targeting claudin 18.2 has shown that it can precisely detect and effectively treat gastric and pancreatic tumours, completely eradicating cancer in some preclinical models.
A newly developed radiopharmaceutical theragnostic pair has demonstrated the ability to precisely detect and effectively treat gastric and pancreatic tumours, completely eradicating cancers in certain preclinical models. The approach targets the biomarker claudin 18.2 and could improve treatment options for some of the most difficult-to-treat solid tumours.
The research, by the Society of Nuclear Medicine and Molecular Imaging, highlights the potential of combining molecular imaging and targeted radiotherapy to move closer to durable disease control – and possibly cure – in upper gastrointestinal cancers.
Major unmet need in upper gastrointestinal cancers
Advanced cancers of the oesophagus, stomach and pancreas are among the deadliest malignancies worldwide, collectively accounting for approximately one-third of all cancer deaths. Prognosis is particularly poor in advanced disease, where treatment options are limited and existing therapies deliver only modest survival benefits.
As a result, there is an urgent need for more precise and effective therapeutic strategies that can both identify suitable patients and directly target cancer cells while avoiding healthy tissue.
Targeting claudin-18.2
Claudin 18.2 has emerged as a promising biomarker in gastric and pancreatic cancers due to its well-defined expression on tumour cells and accessibility for targeted therapies.
Claudin 18.2 recently gained clinical attention following US Food and Drug Administration approval of the gastric cancer treatment zolbetuximab.
“Claudin 18.2 recently gained clinical attention following US Food and Drug Administration approval of the gastric cancer treatment zolbetuximab, which targets the biomarker,” said Shadi Esfahani, MD, MPH, nuclear medicine physician at Massachusetts General Hospital in Boston. “To further explore the utility of claudin 18.2, my colleagues and I developed a first-in-class claudin 18.2-targeted PET radiopharmaceutical and a therapeutic counterpart to identify and treat gastric and pancreatic tumours.”
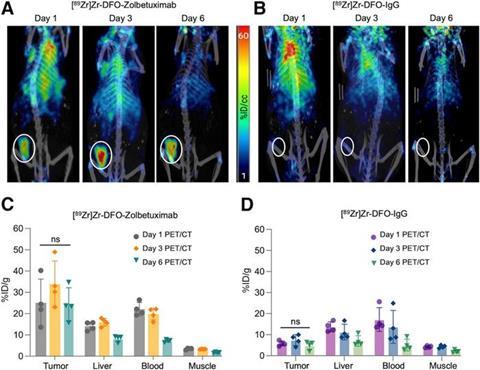
 Evaluation of [89Zr]Zr-DFO-zolbetuximab PET specificity in mouse models of GCa over multiple time points. (A and B) Maximum-intensity-projection serial PET/CT images for male nude mice implanted with GSU gastric tumours (white circles) imaged with [89Zr]Zr-DFO-zolbetuximab or [89Zr]Zr-DFO-IgG 1, 3, and 6 d after injection. (C and D) Quantitative in vivo biodistribution of [89Zr]Zr-DFO-zolbetuximab and [89Zr]Zr-DFO-IgG in tumour and selected tissues. ns = not significant on one-way ANOVA test. Credit: Image created by Dr. S Raheem and Dr. S Esfahani, et al., Massachusetts General Hospital, Boston, MA, USA.[/caption]
Evaluation of [89Zr]Zr-DFO-zolbetuximab PET specificity in mouse models of GCa over multiple time points. (A and B) Maximum-intensity-projection serial PET/CT images for male nude mice implanted with GSU gastric tumours (white circles) imaged with [89Zr]Zr-DFO-zolbetuximab or [89Zr]Zr-DFO-IgG 1, 3, and 6 d after injection. (C and D) Quantitative in vivo biodistribution of [89Zr]Zr-DFO-zolbetuximab and [89Zr]Zr-DFO-IgG in tumour and selected tissues. ns = not significant on one-way ANOVA test. Credit: Image created by Dr. S Raheem and Dr. S Esfahani, et al., Massachusetts General Hospital, Boston, MA, USA.[/caption] Preclinical evaluation shows strong results
To assess the theragnostic pair, researchers developed preclinical murine models using both pancreatic and gastric cancer cell lines. Serial PET imaging was performed using 89Zr-DFO-zolbetuximab, with a control radiotracer used for comparison, followed by detailed biodistribution analysis.
The therapeutic phase involved treating tumour-bearing mice with varying doses of 177Lu-DOTA-zolbetuximab or control agents.
The therapeutic phase involved treating tumour-bearing mice with varying doses of 177Lu-DOTA-zolbetuximab or control agents. Tumour response and potential toxicity were assessed using laboratory testing and histological analysis.
PET imaging demonstrated consistently high tumour uptake of the claudin 18.2-targeted radiopharmaceutical at all measured time points, significantly outperforming the control. High-dose therapeutic treatment led to reduced tumour growth in both gastric and pancreatic cancer models, with most pancreatic tumours showing complete regression. Most importantly, no radiation-induced toxicities were observed during the study.
Implications for patient care
“Claudin 18.2-based theragnostics could meaningfully change patient care in two important ways,” said Esfahani. “First, claudin 18.2-targeted PET imaging enables non-invasive identification of patients whose tumours strongly express this target. Second, claudin 18.2-targeted radiopharmaceutical therapy has the potential to deliver highly focused radiation directly to tumour cells, leading to significant tumour shrinkage and the possibility of improved survival.”
She continued, “More broadly, the study underscores how advances in biomarker discovery, radiopharmaceutical development and patient selection are bringing the field closer to treatments with curative potential rather than incremental benefit.”
While further clinical studies are needed, the results raise the prospect of more effective, personalised therapies – and potentially durable disease control – for patients facing some of the deadliest solid tumours.
Topics
- Biomarkers
- Biopharmaceuticals
- Biotherapeutics
- Cancer
- Disease Research
- Diseases
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Targets
- High-Throughput Screening (HTS)
- Imaging & Diagnostics
- In Vivo Testing
- Massachusetts General Hospital in Boston
- Monoclonal Antibodies
- Neurological disorders
- Oncology
- Shadi Esfahani (nuclear medicine physician at Massachusetts General Hospital in Boston)
- the Society of Nuclear Medicine and Molecular Imaging
- Translational Science