Patient-derived xenograft models are reshaping colorectal cancer research by preserving the complexity of real tumours, potentially helping scientists to develop new therapies in the future.
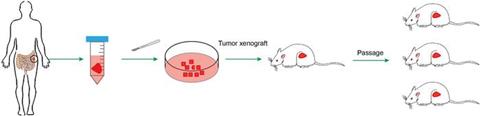
Patient-derived xenograft (PDX) models are rapidly becoming one of the most important tools in colorectal cancer (CRC) research, offering scientists a new view into the behaviour, treatment response and evolution of human tumours. Developed by transplanting fresh tumour tissue from a patient directly into immunodeficient mice, these models retain the original cancer’s genetic, molecular and structural characteristics. As a result, they are increasingly viewed as essential resources in the pursuit of precision oncology.
A complex disease in need of better tools
Colorectal cancer is the third most common cancer globally and remains a major cause of cancer-related deaths. Whilst the number of therapeutic options have increased in recent years, CRC’s extensive genetic diversity and ability to adapt to treatment continue to limit long-term survival outcomes.
Colorectal cancer is the third most common cancer globally and remains a major cause of cancer-related deaths.
Traditional laboratory models, particularly long-established cell lines, have often struggled to recreate the full complexity of human disease. Their limited ability to reproduce tumour heterogeneity has often led to discrepancies between preclinical findings and eventual clinical results.
PDX models, however, offer an advantage. By preserving the tumour microenvironment (TME) and maintaining the diversity of the original cancer cell population, they reflect the intricate biological and pathological traits of individual patients’ tumours. This high level of fidelity makes them a powerful tool for drug discovery, biomarker development and translational research.
Transforming drug testing and personalised medicine
One of the most promising applications of PDX models is their ability to support drug efficacy testing. By analysing how different genetic profiles are linked to responses to specific therapies, researchers can better predict how an individual patient might respond to treatment. This approach aligns closely with the goals of precision medicine, enabling more tailored and targeted therapeutic strategies for CRC patients.
One of the most promising applications of PDX models is their ability to support drug efficacy testing.
PDX models are also key in discovering the mechanisms behind drug resistance. As many patients eventually develop resistance to treatments that initially prove effective, understanding this process has become a critical research priority. Long-term studies using PDX systems have already helped identify molecular pathways that allow cancer cells to evade therapy, providing vital insights that could help researchers develop new next-generation treatments.

Schematic overview of CRC PDX model establishment and propagation in immunodeficient mice. Credit: Genes & Diseases[/caption]
Challenges in establishing and maintaining PDX models
Despite their growing importance, PDX models come with practical challenges. Establishing them is a time-intensive and labour-heavy process that requires high-quality tumour samples, precise engraftment techniques and extensive monitoring. Genetic drift, which can occur over multiple passages, also poses difficulties, potentially changing the tumour’s original features and complicating long-term research.
Despite their growing importance, PDX models come with practical challenges.
Despite these challenges, ongoing advancements – particularly in humanised mouse models designed to better mimic human immune responses – are helping to address these obstacles. It is hoped that these innovations can expand the relevance of PDX models in immunotherapy research, a field of increasing importance in CRC treatment.
A promising future for colorectal cancer research
Looking ahead, experts predict that PDX models will play a crucial and expanding role in co-clinical trials, where laboratory testing is conducted in parallel with patient treatment. By closely mirroring real-world patient outcomes, these models are driving the development of more effective therapeutic strategies and deepening researchers’ understanding of tumour evolution.
As PDX models continue to evolve, they could help to change both the study and treatment of colorectal cancer – offering renewed optimism for improved patient outcomes in the future.




















