A commonly prescribed cholesterol-lowering drug, called pitavastatin, could be used to treat patients with triple-negative breast cancer, after researchers found that it has the ability to block a key cancer survival protein.
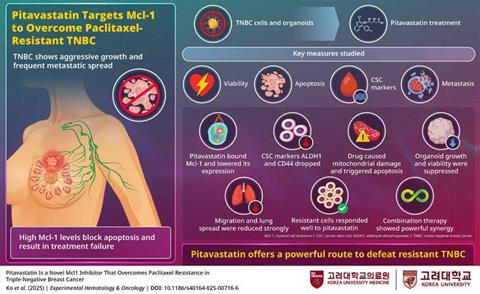
A new study by researchers at Korea University has identified a widely prescribed cholesterol-lowering drug as a potential new treatment for triple-negative breast cancer (TNBC), one of the most aggressive and difficult-to-treat forms of the disease.
TNBC lacks oestrogen, progesterone and HER2 receptors, leaving patients with limited treatment options beyond cytotoxic chemotherapy. While many tumours initially respond to treatment, relapse is common and often rapid, driven by chemotherapy-resistant cancer stem-like cells that seed metastasis.
Targeting a key survival protein
A research team led by Professor Jae Hong Seo at Korea University has discovered that pitavastatin, a statin routinely used to treat high cholesterol, directly inhibits Mcl-1, an anti-apoptotic protein that plays a central role in TNBC cell survival.
Pitavastatin, a statin routinely used to treat high cholesterol, directly inhibits Mcl-1.
The findings, according to Professor Seo, reveal a previously unknown mechanism of action for the drug.
“We report for the first time that pitavastatin is a direct inhibitor of Mcl-1 and targets heterogeneity in TNBC cells via the suppression of CSC-like properties, thereby preventing distant metastasis and counteracting paclitaxel resistance,” he said.
Destroying cancer stem-like cells
Using molecular docking studies and biophysical assays, the researchers showed that pitavastatin binds to the BH3-binding groove of Mcl-1, destabilising the protein and triggering mitochondrial dysfunction. This process led to increased reactive oxygen species production, membrane disruption, cytochrome c release and activation of cell-death pathways.
The drug proved highly effective at eliminating cancer stem-like cell populations. It reduced ALDH1 activity, suppressed CD44^high/CD24^low and CD24^high/CD49^high subpopulations, and sharply inhibited mammosphere formation. Similar effects were observed in patient-derived TNBC organoids, where pitavastatin significantly reduced both size and viability.
 Pitavastatin binds and suppresses Mcl-1, targets cancer stem-like properties, triggers mitochondrial apoptosis, and restores paclitaxel sensitivity in triple-negative breast cancer models. Credit: Professor Jae Hong Seo from Korea University.[/caption]
Pitavastatin binds and suppresses Mcl-1, targets cancer stem-like properties, triggers mitochondrial apoptosis, and restores paclitaxel sensitivity in triple-negative breast cancer models. Credit: Professor Jae Hong Seo from Korea University.[/caption] Strong results in animal models
In cancer stem cell-derived allograft mouse models, pitavastatin reduced tumour growth, angiogenesis and lung metastasis. Importantly, these anti-tumour effects occurred without evidence of organ toxicity or significant body weight loss.
Tumour tissue analysis also demonstrated a substantial reduction in the proliferation marker Ki-67 alongside increased apoptosis and suppressed blood vessel formation. Circulating levels of metastasis-associated factors, including MMP-2, MMP-9 and VEGF, were also significantly lowered.
Overcoming chemotherapy resistance
Paclitaxel-resistant TNBC cells, which typically exhibit elevated Mcl-1 levels, enhanced stemness and activation of JAK2–STAT3 signalling, remained highly sensitive to pitavastatin. The drug downregulated Mcl-1 and Bcl-2, reduced MDR1/P-gp expression, suppressed STAT3 activation and restored mitochondrial apoptosis.
Our results support pitavastatin as a promising candidate for drug repurposing.
When combined with paclitaxel, pitavastatin synergistically inhibited TNBC organoid growth, outperforming either treatment alone. In resistant mammospheres, it eliminated cancer stem cell frequency and prevented metastatic lung colonisation in vivo.
“Our results support pitavastatin as a promising candidate for drug repurposing, particularly in TNBC characterised by high Mcl-1 expression and resistance to conventional chemotherapy,” said Professor Seo.
A pathway to faster clinical impact
The study highlights the potential of drug repurposing as a strategy to accelerate new cancer treatments. With its established safety profile and mechanism-based targeting of Mcl-1, pitavastatin could offer a faster route to clinical translation for patients with chemotherapy-refractory TNBC, a group for whom new therapeutic options are urgently needed.