Scripps Research scientists have discovered that post-translational modifications significantly influence whether proteins can be targeted by drugs, identifying over 400 proteins whose druggability depends on their chemical modification state.

Scientists have discovered a factor that could modify the way in which new medicines are developed, after discovering that subtle chemical changes to proteins can determine whether they respond to drugs.
The research, led by Scripps Research, shows that post-translational modifications, or PTMs, play a major role in controlling how proteins interact with drug-like molecules.
Chemical changes influence drug targeting
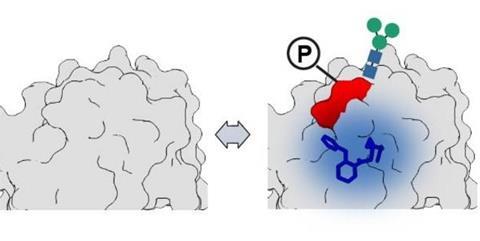
Inside human cells, proteins are routinely altered after they are produced through PTMs, small chemical modifications that can affect how proteins fold, move and function. The new study reveals that these modifications also influence whether proteins can be effectively targeted by drugs.
Researchers identified more than 400 proteins whose ability to bind drug-like molecules depends on their modification state. This suggests that a single protein may be more or less ’druggable’ depending on which chemical tags it carries at a given time.
Inside human cells, proteins are routinely altered after they are produced through PTMs, small chemical modifications that can affect how proteins fold, move and function
“We already know PTMs affect protein structure and function,” said Christopher Parker, a professor and The Abide-Vividion Chair in Chemistry and Chemical Biology at Scripps Research and a senior author of the study. “We asked: if we perturb a class of PTMs, what’s the broad impact on proteome-wide druggability?”
Mapping drug access across the proteome
To investigate, the team used specially designed chemical probes, small molecules that can attach permanently to the proteins they bind. These probes were applied in living cells under conditions that altered two common types of PTMs: phosphorylation and N-linked glycosylation.
This approach allowed scientists to track how changes in PTMs affected the accessibility of proteins to drug-like molecules across more than 5,000 targets.
The results showed that there was a widespread impact, spanning enzymes, ion channels, transcription factors and epigenetic regulators, including many proteins that have historically proved difficult to target with drugs.
In some cases, PTMs were located close to drug binding sites, directly modifying the pockets where molecules attach. In others, they altered how proteins interact with each other, revealing entirely new binding opportunities.
Implications for cancer therapies
One of the most significant findings involved KRAS, a protein frequently mutated in cancer and a key target for modern therapies. Drugs such as sotorasib and adagrasib are already used to treat certain forms of non-small cell lung cancer.
The researchers found that phosphorylation at specific sites on KRAS can significantly influence how effectively these inhibitors bind and suppress the protein.
One of the most significant findings involved KRAS, a protein frequently mutated in cancer and a key target for modern therapies
This discovery may help explain why such treatments vary in effectiveness between patients and suggests that a tumour’s PTM profile could play a role in guiding therapy decisions.
“Even with a small searchlight we found that one small PTM on one site dramatically affects the activity of known inhibitors for one of the most sought-after drug targets in cancer,” said Parker. “Those inhibitors work well in patients but maybe they could work better.”
Beyond cancer: wider disease relevance
The findings aren’t just important for oncology. Among the proteins identified was NPC2, which is linked to Niemann-Pick disease, a rare and often fatal condition. In this case, a single sugar-based PTM determined whether drug-like molecules could bind to the protein.
“PTMs are another layer of variables that we should be considering more in drug discovery,” said Parker.
Towards more precise medicines
The study suggests that understanding PTM states could enable researchers to design more selective therapies with fewer unintended effects. Many of the proteins identified currently lack effective drug candidates, highlighting potential new avenues for treatment.
The study suggests that understanding PTM states could enable researchers to design more selective therapies with fewer unintended effects
Looking ahead, the team plans to expand its approach to other types of PTMs and more disease-relevant systems, using larger and more diverse chemical libraries.
“Our vision would be to broadly achieve disease-state-specific pharmacology,” said Parker. “PTM status could be a way to selectively target a protein in a disease cell, finding vulnerabilities like unique chinks in the armor of a disease.”
















No comments yet