Researchers have discovered how to inhibit the P2X4 receptor, a key protein linked to chronic pain, inflammation and certain cancers.
Scientists at the University of Bonn and University Hospital Bonn (UKB) have discovered a molecular mechanism that could allow for the development of new treatments targeting chronic pain, inflammation and certain cancers. Their findings reveal how a promising compound can inhibit the human P2X4 receptor – a protein central to several hard-to-treat conditions.
A receptor linked to pain, inflammation and cancer
The P2X4 receptor sits within the membranes of many cell types, acting as a gated channel that responds specifically to ATP, a molecule known as the body’s universal energy currency. Once ATP binds, the receptor opens and allows sodium and calcium ions to enter the cell, triggering a range of physiological responses.
In immune cells, this activation can spark inflammation, whilst in certain nerve cells, it can generate persistent pain signals. Some tumour cells also exploit this pathway, using ATP-driven P2X4 activity to fuel their growth and spread.
“This receptor is often overactive in conditions such as chronic inflammation or chronic pain,” explains Dr Christa Müller, Head of Pharmaceutical & Medicinal Chemistry at the University of Bonn. “The same is true of some tumour cells – which are driven to keep dividing by ATP and can thus also form metastases.”
Pharmaceutical companies have long searched for molecules able to block or dampen the receptor’s activity, but with limited success. One promising candidate is PSB-0704, an anthraquinone derivative developed by Müller’s team. “We wanted to find out what it actually does and at the same time use this knowledge to help in the development of better drugs,” she said.
Cryo-EM reveals the binding mechanism
Understanding how PSB-0704 interacts with P2X4 proved challenging, as earlier attempts to crystallise the complex were unsuccessful. The research team turned instead to cryogenic electron microscopy (cryo-EM), enabling them to visualise millions of frozen receptors–inhibitor complexes at near-atomic resolution.
Understanding how PSB-0704 interacts with P2X4 proved challenging, as earlier attempts to crystallise the complex were unsuccessful.
“For this method, we produced a solution of the P2X4 receptor and the anthraquinone derivative PSB-0704 and then snap froze it,” said lead author Dr Jessica Nagel. “The resulting film of ice contains millions of receptor molecules together with the bound inhibitor, which we can examine under an electron microscope.”
Working closely with Dr Gregor Hagelüken at UKB’s Institute of Structural Biology, the team reconstructed detailed 3D models of the receptor.
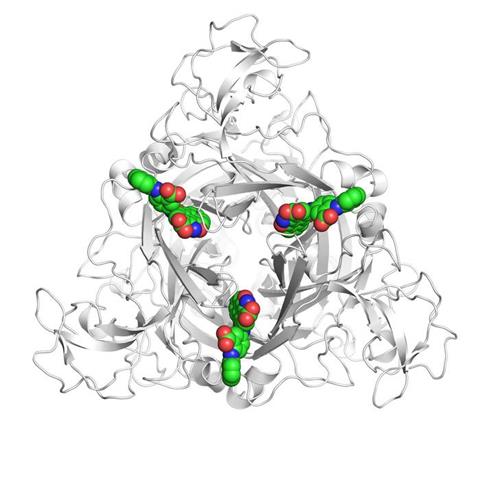
Their analysis revealed that PSB-0704 prevents the ion channel from opening by shifting parts of the P2X4 structure. “When the inhibitor bonds, it causes parts of the P2X4 molecule to move so that it is no longer possible to open the ion channel,” Nagel explains. The channel remains locked even when ATP is present.
 (seen here from above) has three parts. If activated, a pore in the receptor opens and allows ions to flow into the cell. PSB-0704 (shown in colour) prevents this from happening and blocks, for example, the transmission of pain signals. Credit: AG Müller/Hagelüken/University of Bonn.[/caption]
(seen here from above) has three parts. If activated, a pore in the receptor opens and allows ions to flow into the cell. PSB-0704 (shown in colour) prevents this from happening and blocks, for example, the transmission of pain signals. Credit: AG Müller/Hagelüken/University of Bonn.[/caption] A ‘rubber band’ limiting drug effectiveness
Although PSB-0704 can inhibit P2X4, it does so only at relatively high concentrations. The researchers discovered that the compound binds within a small pocket that it does not fit particularly well within.
This pocket is constrained by what Nagel describes as a molecular ‘rubber band’. “We have developed a receptor without this rubber band,” she said. “And the PSB-0704 inhibitor was almost 700 times more potent as a result.”
Towards better-targeted therapies
The findings provide crucial insights for designing future drugs. “On the one hand, we can try to design drugs that cut through the molecular rubber band before they bind with the P2X4 receptor,” Müller notes. “An alternative would be to search for smaller molecules that can fit more easily into the binding pocket.”
Müller cautions that there is still some way to go, but emphasises that the study has now given them a strong base upon which they could successfully achieve their goal of developing new drugs that can effectively inhibit the opening of the P2X4 receptor.
Topics
- Autoimmune disease
- Cancer
- Cancer
- Dr Christa Müller (Head of Pharmaceutical & Medicinal Chemistry at the University of Bonn)
- Drug Development
- Drug Discovery Processes
- Drug Targets
- High-Throughput Screening (HTS)
- Hit-to-Lead
- Ion Channels
- Microscopy
- Molecular Biology
- Neurons
- Oncology
- Pharmacology
- Rare & Genetic Disorders
- Small Molecules
- Structural Biology
- the University of Bonn
- Translational Science
- University Hospital Bonn (UKB)