Scientists have identified a little-known protein, FGD3, that can boost the power of key breast cancer treatments by helping drugs rupture tumour cells and activate the immune system.
A naturally occurring protein found at elevated levels in breast cancer cells could significantly improve the effectiveness of key anticancer therapies, according to new research. Scientists at the University of Illinois report that the protein, FGD3, enhances the impact of widely used drugs such as doxorubicin, as well as the experimental agent ErSO, by helping to rupture cancer cells and stimulate immune responses. This discovery could strengthen chemotherapy and immunotherapy approaches for breast cancer in the future.
A promising lead from experimental drug ErSO
The discovery emerged from investigations into ErSO, an experimental drug previously shown to kill 95-100 percent of oestrogen-receptor-positive breast cancer cells in a mouse model. ErSO acts by overactivating a cellular stress-response pathway that cancer cells usually rely on for protection.
The discovery emerged from investigations into ErSO, an experimental drug previously shown to kill 95-100 percent of oestrogen-receptor-positive breast cancer cells in a mouse model.
“Most anticancer drugs inhibit something that the cell needs to survive, and they either prevent the cell from growing or, in some cases, cause it to die in an orderly way called apoptosis,” said University of Illinois Urbana-Champaign biochemistry professor David Shapiro, who led the study with graduate student Junyao Zhu. “But ErSO does exactly the opposite. It over activates the cell pathway and the cancer cells literally swell up and rip open.”
Shapiro, along with chemistry professor Paul Hergenrother, first identified ErSO in 2021. The new study aimed to clarify how the drug functions by identifying proteins involved in ‘making life-death decisions for the cells’.
Discovering the role of FGD3
To pinpoint these proteins, the team screened breast cancer cell lines in which individual genes had been deleted. If the absence of a gene weakened ErSO’s effect, it suggested that the corresponding protein played a role in the drug’s action.
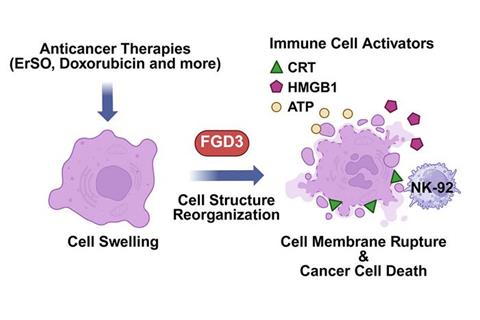
“The top target from the screen with ErSO was the gene for this little-studied protein called FGD3,” Shapiro said. “So, we manipulated levels of FGD3 in cancer cells and saw that it indeed controlled whether ErSO could kill the cells. And in a series of important experiments, Zhu showed that FGD3 weakens the cell’s architecture.”
Under normal conditions, FGD3 increases cell flexibility, enabling cancer cells to move and potentially metastasise. However, when exposed to ErSO or doxorubicin, the protein contributes to the cells’ rupture. This rupture releases cell contents, triggering an immune response in which natural killer cells and macrophages move in to destroy the damaged cancer cells.
 Anticancer drugs cause breast cancer cells to swell. The cellular protein, FGD3, alters the disrupted cancer cells, promoting the display of molecules on their surface that recruit immune cells, researchers found. The swollen cancer cells are more likely to burst in the presence of high levels of FGD3. Credit: graphic created in BioRender. Zhu, J. (2025) https://BioRender.com/wq32727[/caption]
Anticancer drugs cause breast cancer cells to swell. The cellular protein, FGD3, alters the disrupted cancer cells, promoting the display of molecules on their surface that recruit immune cells, researchers found. The swollen cancer cells are more likely to burst in the presence of high levels of FGD3. Credit: graphic created in BioRender. Zhu, J. (2025) https://BioRender.com/wq32727[/caption] Validating findings in advanced models
The researchers tested the mechanism in both traditional 2D cell cultures and 3D patient-derived breast cancer organoids, which closely replicate the protein expression profiles of actual tumours.
The researchers tested the mechanism in both traditional 2D cell cultures and 3D patient-derived breast cancer organoids.
The team also confirmed the effect of FGD3 in a mouse model, observing that higher levels of the protein boosted ErSO’s tumour-killing capability. “One of the things we saw was that FGD3 dramatically increased the movement to the cancer cell membrane of a protein that stimulates natural killer cells to target a cancer cell for destruction,” said Shapiro.
Potential for better targeted therapies
Analysis of extensive human breast cancer datasets demonstrated strong correlations between FGD3 levels and patient responses to chemotherapy. “Those with a high level are highly responsive; those with a low level are poorly responsive,” Shapiro said. This insight could help clinicians in identifying patients most likely to benefit from specific treatments.
Looking towards the future, the researchers say they will try to expand FGD3 into a wider context, to show whether it also plays a role in other cancers and cancer therapies.
Topics
- 3D Cell Cultures
- Analytical Techniques
- Cancer
- Cancer
- Chemotherapy
- David Shapiro (University of Illinois Urbana-Champaign biochemistry professor)
- Drug Discovery
- Drug Discovery Processes
- Drug Targets
- Immuno-oncology
- Immuno-oncology
- Immunotherapy
- In Vivo Testing
- Molecular Biology
- Oncology
- Organoids
- Tools and techniques
- Translational Science
- University of Illinois




















