Researchers have engineered new extracellular vesicles that can selectively induce antigen-specific regulatory T cells, possibly leading towards safer, more precise treatments for autoimmune and allergic diseases.
Researchers at the Nano Life Science Institute (WPI-NanoLSI) and the Faculty of Medicine at Kanazawa University have developed a new class of engineered extracellular vesicles that can induce antigen-specific regulatory T cells, a key component of the immune system responsible for restraining excessive or misdirected immune responses. The work could help in the development of a new generation of targeted therapies for autoimmune and allergic diseases.
Autoimmune conditions arise when the immune system mistakenly attacks the body’s own tissues. Current treatments rely heavily on steroids or broad immunosuppressive drugs, which can reduce symptoms but also weaken normal immune defences, increasing the risk of infection and other serious complications. For decades, immunologists have sought ways to suppress immune reactions only against disease-related targets, a strategy known as antigen-specific immune tolerance.
Harnessing the body’s natural immune regulators
Regulatory T cells, or Tregs, play a central role in maintaining immune tolerance and preventing harmful immune activation. However, inducing antigen-specific Tregs safely and efficiently inside the body has remained a major challenge.
To overcome this, the researchers, led by Shota Imai, Tomoyoshi Yamano and Rikinari Hanayama, engineered what they call antigen-presenting extracellular vesicles, or AP-EVs-Treg. These vesicles are designed to display three critical components on a single surface: peptide–MHC class II complexes that allow antigen-specific recognition by T cells, and the cytokines interleukin-2 and transforming growth factor-β, both essential for Treg differentiation.
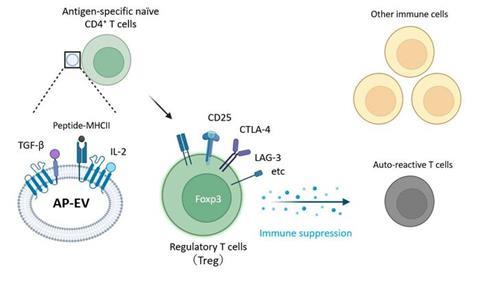
 Engineered antigen-presenting extracellular vesicles (AP-EVs) deliver peptide-MHCII, IL-2, and TGF-β to naïve antigen-specific CD4⁺ T cells, driving their differentiation into Foxp3⁺ regulatory T cells (Tregs). The induced Tregs suppress autoreactive T cells and modulate surrounding immune cells to promote antigen-specific immune tolerance. Credit: © Tomoyoshi Yamano.[/caption]
Engineered antigen-presenting extracellular vesicles (AP-EVs) deliver peptide-MHCII, IL-2, and TGF-β to naïve antigen-specific CD4⁺ T cells, driving their differentiation into Foxp3⁺ regulatory T cells (Tregs). The induced Tregs suppress autoreactive T cells and modulate surrounding immune cells to promote antigen-specific immune tolerance. Credit: © Tomoyoshi Yamano.[/caption] Robust induction of functional Tregs in the laboratory
When the engineered vesicles were co-cultured with naïve CD4⁺ T cells from antigen-specific T-cell receptor transgenic mice, they efficiently drove the formation and expansion of Foxp3⁺ regulatory T cells. The induced cells expressed high levels of suppressive molecules, including CTLA-4, PD-L1 and LAG-3, and strongly inhibited the proliferation of other T cells in a dose-dependent manner, demonstrating potent immune-suppressive function.
Crucially, the system was adaptable. By changing the peptide loaded onto the vesicles, the researchers were able to generate Tregs specific to different disease-related antigens, including myelin oligodendrocyte glycoprotein peptides associated with multiple sclerosis.
Synergistic effects in animal models
In vivo experiments showed that AP-EVs selectively activated antigen-specific CD4⁺ T cells according to their peptide–MHC specificity. However, the full induction of Foxp3⁺ Tregs required co-administration of rapamycin, an mTOR inhibitor already known to favour Treg differentiation. Together, AP-EVs and rapamycin markedly increased the generation of antigen-specific regulatory T cells, revealing a synergistic strategy for restoring immune tolerance in living systems.
A flexible and clinically promising platform
Extracellular vesicles offer several advantages over other tolerogenic approaches, such as mRNA or synthetic nanoparticle systems. They are naturally derived, highly biocompatible and have low immunogenicity, while being capable of presenting multiple functional molecules at once. The modular design of the AP-EV platform allows both antigen specificity and immunoregulatory signals to be tuned, supporting future applications across a wide range of autoimmune and allergic diseases.
With more than 80 autoimmune disorders affecting hundreds of millions of people worldwide, the development of a precise, antigen-specific immune-tolerance technology could represent a significant advance in long-term, safer treatment strategies.
Topics
- Allergic diseases
- Autoimmune disease
- Biologics
- Biotherapeutics
- Cell & Gene Therapy
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Targets
- Immunology
- Immunotherapy
- In Vitro Testing
- In Vivo Testing
- Molecular Biology
- Nanoparticles
- Nanotechnology
- Neurological disorders
- T Cells
- the Faculty of Medicine at Kanazawa University
- the Nano Life Science Institute (WPI-NanoLSI)
- Translational Science