Scientists in Japan have identified a way to weaken the dense fibrotic barrier that prevents drugs from penetrating pancreatic tumours – a promising new strategy that could improve treatment for pancreatic cancer.
Pancreatic cancer is one of the most lethal forms of the disease, with survival rates very low, despite decades of studies in oncology. A major obstacle to treatment has been the disease’s dense and fibrotic microenvironment – a collagen-heavy tissue structure that forms both a physical and biochemical blockade, preventing drugs from adequately reaching tumour cells.
A new collaborative study from Okayama University and Tohoku University now suggests that it is possible to overcome this barrier.
Researchers identify a key signalling pathway behind fibrotic resistance
A team led by Assistant Professor Hiroyoshi Y. Tanaka from the Graduate School of Medicine, Dentistry and Pharmaceutical Sciences at Okayama University has discovered that blocking collagen signalling via the DDR1 receptor significantly improves the delivery of macromolecular drugs to pancreatic tumours.
Our findings reveal that collagen signalling, not just its physical density, plays a crucial role in hindering drug delivery.
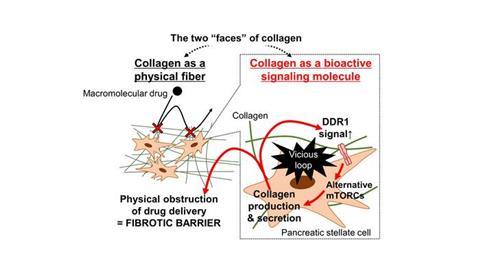
The findings present a novel therapeutic pathway aimed at weakening the fibrotic defences that shield cancer cells. The research then examined how collagen – typically regarded only as a structural obstacle – also acts as an active signalling molecule influencing fibrosis and drug permeability.
“Our findings reveal that collagen signalling, not just its physical density, plays a crucial role in hindering drug delivery,” explained Dr Tanaka, a co-author from Okayama University. “By inhibiting DDR1, we can interrupt this signalling cascade, loosen the fibrotic barrier and enable better access for therapeutic agents.”
Advanced 3D models reveal how DDR1 inhibition boosts drug penetration
Using an advanced 3D cell model that mimics the fibrotic environment of pancreatic cancer, the team showed that blocking DDR1 signalling reduces fibrosis and improves the movement of large therapeutic molecules such as antibodies and nanomedicines.
This breakthrough suggests that DDR1 inhibition could play a key role in enabling next-generation treatments to work more effectively in one of oncology’s most drug-resistant cancers.
 Collagen plays dual roles in pancreatic cancer: as a physical fiber that obstructs macromolecular drug delivery, and as a bioactive signaling molecule that activates discoidin domain receptor 1 (DDR1) and drives a vicious fibrotic cycle. By blocking DDR1 signaling, researchers from Okayama and Tohoku Universities demonstrated improved drug penetration and reduced fibrosis, uncovering a new pathway for therapeutic intervention. Credit: Dr. Hiroyoshi Y. Tanaka and Professor Mitsunobu R. Kano from Okayama University, Japan.[/caption]
Collagen plays dual roles in pancreatic cancer: as a physical fiber that obstructs macromolecular drug delivery, and as a bioactive signaling molecule that activates discoidin domain receptor 1 (DDR1) and drives a vicious fibrotic cycle. By blocking DDR1 signaling, researchers from Okayama and Tohoku Universities demonstrated improved drug penetration and reduced fibrosis, uncovering a new pathway for therapeutic intervention. Credit: Dr. Hiroyoshi Y. Tanaka and Professor Mitsunobu R. Kano from Okayama University, Japan.[/caption] MEK inhibitors linked to unexpected increase in fibrosis
One of the study’s most interesting discoveries was that MEK inhibitors – a class of drugs previously trialled for pancreatic cancer – appear to intensify the fibrotic barrier. The researchers found that these drugs increase collagen I expression which created an even thicker barrier that hinders drug delivery.
“We found that while MEK inhibitors can attack cancer cells, they also unintentionally strengthen the fibrotic barrier, making drug penetration even more difficult,” said Dr Tanaka. “Recognising and countering this effect could fundamentally change how combination therapies are designed for pancreatic cancer.”
Implications beyond pancreatic cancer
The team emphasises that their findings may extend beyond one disease. A deeper understanding of collagen signalling could lead to new treatment strategies for many different fibrotic conditions in oncology and other medical fields.
A deeper understanding of collagen signalling could lead to new treatment strategies for many different fibrotic conditions.
Looking ahead, the researchers plan to investigate combination therapies that simultaneously target tumour cells and their fibrotic surroundings, with the goal of pushing these insights from laboratory models towards clinical application.
As research continues into pancreatic cancer, this study offers fresh optimism – showing that rethinking the biology of fibrosis may be central to helping life-saving drugs finally reach their intended targets.
Topics
- 3D Cell Cultures
- Analytical Techniques
- Assistant Professor Hiroyoshi Y. Tanaka (Graduate School of Medicine Dentistry and Pharmaceutical Sciences at Okayama University)
- Biologics
- Cancer
- Drug Delivery
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Targets
- Molecular Biology
- Molecular Targets
- Monoclonal Antibodies
- Nanomedicine
- Neurological disorders
- Okayama University
- Oncology
- Organoids
- Tohoku University
- Translational Science