Scientists have restored sperm production in mice with a genetic form of male infertility using mRNA delivered via lipid nanoparticles, with the hope of informing future treatments for non-obstructive azoospermia.
A new study by researchers at The University of Osaka, in collaboration with Baylor College of Medicine, have developed a new treatment for male infertility caused by genetic defects. By delivering mRNA through lipid nanoparticles (LNPs) targeting specific testicular genes, scientists successfully restored sperm production and produced viable offspring in a mouse model.
Male infertility and NOA
Infertility affects one in six couples worldwide, with male factors accounting for almost half of all cases. Among these, non-obstructive azoospermia (NOA) presents a particularly challenging problem. Men with NOA produce no sperm in their ejaculate despite normal hormone levels, often due to genetic defects that disrupt spermatogenesis. Current treatments are limited, leaving thousands of affected men without viable options for conceiving biologically.
A mouse model breakthrough
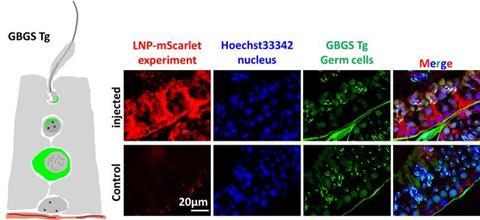
The study focused on a NOA mouse model experiencing meiotic arrest caused by a specific genetic deficiency. Researchers injected LNPs into the rete testis to distribute mRNA throughout the seminiferous tubules. Expression of the mRNA lasted approximately five days and reached around 55 percent of the tubules.
To ensure the mRNA preferentially targeted germ cells rather than Sertoli cells, the team added the Dsc1 3’-UTR containing a miR-471 target. This adjustment shifted translation toward germ cells. In mice lacking the Pdha2 gene, which normally arrests meiosis, delivered Pdha2 mRNA resumed meiotic progression. Within two weeks, round spermatids were observed, followed by fully formed sperm by the third week.
Using these testicular sperm in intracytoplasmic sperm injection (ICSI) procedures, researchers produced 26 pups from 117 embryos, a success rate of 22.2 percent. All offspring developed normally, were fertile and displayed no large genomic alterations greater than 1 megabase – 1 million base pairs (bp) of DNA.

LNP is delivered to both germ cells and Sertoli cells. Credit: Mashiko et al., 2025. Published in PNAS under CC-BY license.[/caption]
A safer alternative to gene therapy
This study demonstrates a new approach for tackling male infertility at the genetic level. Unlike traditional gene therapies, which carry risks of genome integration, LNP-mediated mRNA delivery provides a non-integrating alternative.
This study demonstrates a new approach for tackling male infertility at the genetic level.
Professor Masahito Ikawa, senior author of the study, emphasised the significance of the technique:
“Using fully synthetic LNPs to deliver mRNA minimises genome-integration concerns and enables us to restore spermatogenesis in a defined genetic model.”
Looking ahead
The success of this approach in mice could lead to potential gene-informed treatments for men with NOA, who currently have few options. While further research is needed to adapt the technique safely for human use, the study could one day treat the condition which has long been considered untreatable.
Topics
- Azoospermia
- Baylor College of Medicine
- Cell & Gene Therapy
- Drug Discovery
- Drug Discovery Processes
- Endocrine disorders
- Gene Therapy
- Genomics & Sequencing
- High-Throughput Screening (HTS)
- In Vitro Testing
- In Vivo Testing
- intra-cytoplasmic sperm injection
- Masahito Ikawa (The University of Osaka)
- Molecular Biology
- Nanomedicine
- Nanoparticles
- RNAs
- Stem Cells
- The University of Osaka
- Translational Science