Researchers have identified new drug candidates that selectively target the cPLA2 enzyme, a key driver of brain inflammation linked to Alzheimer’s disease, offering a potential new approach to reducing risk in people with the APOE4 gene.
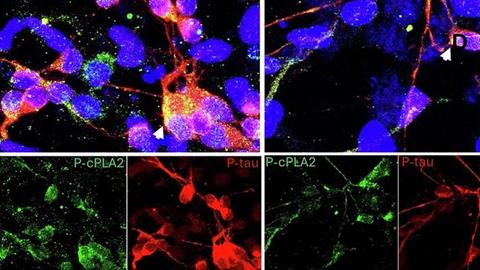
A multidisciplinary research team at the University of Southern California has identified new compounds that could help target a key driver of brain inflammation associated with Alzheimer’s disease. The findings could help to inform new treatments that address inflammation without disrupting normal brain function.
The research focused on an enzyme known as calcium-dependent phospholipase A2 (cPLA2) which plays a central role in inflammatory processes in the brain. The team discovered its significance by studying individuals who carry the APOE4 gene, the strongest known genetic risk factor for Alzheimer’s disease.
While many people with the APOE4 gene never develop Alzheimer’s, the researchers found that those with elevated levels of cPLA2 are far more likely to do so. This connection highlights cPLA2 as a potential therapeutic target but also presents a challenge, as the enzyme is essential for healthy brain activity.
Targeting inflammation without harming the brain
Because cPLA2 is involved in normal brain function, any drug designed to inhibit it must be precise. A successful compound would need to reduce excessive enzyme activity without shutting it down entirely. It must also be small enough to cross the blood–brain barrier, a major hurdle in developing effective neurological treatments.
Because cPLA2 is involved in normal brain function, any drug designed to inhibit it must be precise.
“In this study, we identified compounds that act selectively on cPLA2, with minimal effects on related PLA2 enzymes that are important for normal cellular function,” said senior author Hussein Yassine, Director of the Center for Personalized Brain Health at the Keck School of Medicine of USC. “Across cell-based and animal models, cPLA2 activity was reduced at low concentrations, indicating that the compounds are potent in brain-relevant systems.”
 The team evaluated billions of potential molecules, prioritising those predicted to be selective, brain-penetrant and active under biologically relevant conditions. Credit: Image/USC.[/caption]
The team evaluated billions of potential molecules, prioritising those predicted to be selective, brain-penetrant and active under biologically relevant conditions. Credit: Image/USC.[/caption]Screening billions of molecules
To find suitable candidates, the team employed large-scale computational screening methods to analyse billions of potential molecules. The aim was to prioritise compounds predicted to be selective, able to penetrate the brain and active under biologically relevant conditions.
To find suitable candidates, the team employed large-scale computational screening methods to analyse billions of potential molecules.
The screening techniques were developed by Vsevolod “Seva” Katritch of the USC Dornsife College of Letters, Arts and Sciences and the USC Michelson Center for Convergent Bioscience. This approach allowed researchers to narrow down large chemical possibilities to a manageable group of promising candidates.
Once these top candidates were identified, pharmacologist Stan Louie of the USC Alfred E. Mann School of Pharmacy and Pharmaceutical Sciences led efforts to prepare the compounds for testing. His team assessed how effectively the molecules could be delivered and measured their levels in the brain of animal models.
Encouraging results in laboratory and animal studies
One cPLA2 inhibitor emerged as the leading candidate after it successfully reduced pathological cPLA2 activation in human brain cells exposed to Alzheimer’s-related stressors. Further testing in mouse models showed that the inhibitor could cross the blood–brain barrier and modulate neuroinflammatory pathways.
These results suggest that selectively inhibiting cPLA2 may offer a promising therapeutic strategy, not only for Alzheimer’s disease but potentially for other neurodegenerative conditions linked to inflammation.
“Our goal is to find out whether targeting inflammation can alter Alzheimer’s risk – particularly in APOE4 carriers,” Yassine said. “This next phase focuses not on promises, but on carefully determining whether modulating this pathway is safe, feasible and ultimately meaningful for human disease.”
Topics
- Computational Techniques
- Disease Research
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Leads
- Drug Targets
- Enzymes
- High-Throughput Screening (HTS)
- Hit-to-Lead
- Hussein Yassine (Director of the Center for Personalized Brain Health at the Keck School of Medicine of USC)
- In Vitro Testing
- In Vivo Testing
- Molecular Biology
- Neurological disorders
- Neuroprotection
- Neurosciences
- Pharmacology
- Small Molecules
- Translational Science
- University of Southern California (USC)