A microscopic DNA nanostructure inspired by bacteriophages can deliver therapeutic molecules directly into cells whilst evading endosomal traps that typically render treatments ineffective.
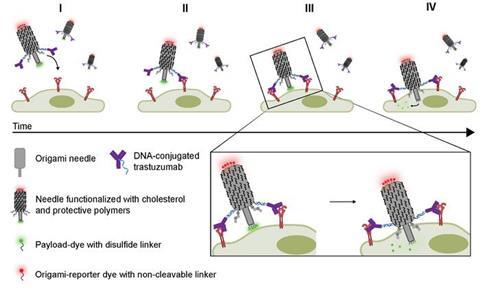
Researchers at Aarhus University have developed a microscopic DNA ’needle’ capable of delivering molecules directly into cells, offering a potential breakthrough in how medicines are transported and activated inside the body.
Tackling a long-standing medical challenge
One of the major obstacles in modern medicine is that substances entering a cell are often trapped inside small compartments and made to be ineffective before reaching their target. These compartments, known as endosomes, act as part of the cell’s defence system.
The newly developed DNA structure aims to overcome this limitation by ensuring that therapeutic molecules not only enter cells but remain active once inside.
Inspired by viruses
The innovation draws inspiration from bacteriophages, viruses that infect bacteria by injecting genetic material into cells. The Danish research team has engineered a synthetic version of this mechanism.
Unlike natural viruses, the artificial bacteriophage carries no genetic material
Unlike natural viruses, the artificial bacteriophage carries no genetic material. Instead, it can be loaded with tailor-made molecules and programmed to target specific cell types, delivering its payload directly into the cell interior.
A possible breakthrough
Most importantly, the DNA needle appears to bypass the cell’s usual defences, meaning the delivered molecules are not trapped in endosomes and are therefore able to function as intended.
In current treatments for rare genetic disorders, cells are often given oligonucleotides, short strands of DNA or RNA delivered via nanoparticles. However, only around one percent of these molecules successfully escape into the cell where they are needed.
Early laboratory success
So far, the researchers have demonstrated the technique in laboratory conditions, successfully delivering a dye payload into breast cancer cells.
So far, the researchers have demonstrated the technique in laboratory conditions, successfully delivering a dye payload into breast cancer cells
“We have not yet succeeded in getting large molecules into the cell with this method,” said Kurt Vesterager Gothelf, Professor at Aarhus University and senior author of the research. ”That is the next big step, and it will be a milestone in determining whether the method can be used medically. Right now, we are actively seeking funding to continue the study.”
Potential beyond cancer
Although breast cancer cells were used in the study for practical reasons, the technology could in principle be applied to a wide range of diseases requiring delivery of proteins or genetic material into cells.
Initially, the researchers expect the method to be expensive, meaning early applications may focus on severe or rare genetic disorders.
“But in the longer term, if it can be produced more cheaply, it could be extended to many other treatments, including cancer,” said Mette Galsgaard Malle, Assistant Professor and a key member of the research team.
How the DNA needle works
The system is built using a technique known as DNA origami, in which strands of DNA are programmed to fold into specific shapes.
The resulting structure functions like a modular delivery platform. Antibodies on its surface bind to target cells, while a cholesterol base anchors it to the cell membrane and helps drive the needle inside. A polymer coating protects the structure and the therapeutic payload is attached to the tip.
The system is built using a technique known as DNA origami, in which strands of DNA are programmed to fold into specific shapes
Once inside the cell the payload is released through a chemical process and can move freely towards its target.
“All of those functional units are important for achieving exactly the function we want,” said Malle. ”If, for example, it does not have the cleavable ends on the part of the needle that enters the cell to deliver the payload, then it does not deliver anything. At the same time, we now have a platform for investigating which units are needed to construct a specific delivery method.”
A long road to clinical use
Despite the promise, there are still challenges ahead. Researchers must demonstrate that the system can deliver larger molecules efficiently and determine whether it could trigger immune reactions or toxicity in humans.
Additional protective layers may also be required before the technology can be used clinically.
While clinical use may still be years away, the DNA needle represents a flexible platform that could be adapted for a wide range of future treatments, potentially transforming how medicines are delivered at the cellular level.














No comments yet