TxCell financial results for the first half of 2014
Posted: 25 September 2014 | TXCELL | No comments yet
TxCell reports its financial results for the first half of 2014…

TxCell (FR0010127662 – TXCL), a biotechnology company developing innovative, personalized cell-based immunotherapies using antigen specific regulatory T-cells (Ag-Tregs) for severe chronic inflammatory and autoimmune diseases, today reports its financial results for the first half of 2014.
The Half Year Financial Report as of June 30, 2014 is available on the Company’s website: www.txcell.com.
The following interim financial statements as of June 30, 2014, established according to IFRS standards, have been subject to a limited review by the Statutory Auditors and have been approved by the Board of Directors of the company on September 22, 2014.
Highlights of TxCell’s results as of June 30, 2014 are:
- The ongoing recognition of the first milestone of EUR 1 million paid by Ferring at the signing of their partnership late 2013;
- A consistent and controlled increase of operating expenses due to the launch of the development of Col-Treg, TxCell’s second product candidate in autoimmune uveitis, and the upcoming start of the phase IIb clinical study with Ovasave(R) in Crohn’s disease;
- A strong financial position with EUR 17.4 million in cash as of June 30, 2014, allowing TxCell to cover its medium-term needs based on its current programs.
Damian Marron, Chief Executive Officer of TxCell, says:
“The first half of 2014 was marked by a number of important events for TxCell. The Initial Public Offering of the company gives financial visibility in the medium term and allows us to accelerate the clinical development of our next-generation cell therapies. Obtaining the GMP certificate for TxCell’s manufacturing platform was a key step in the development of company and especially for our first product candidate Ovasave for which we expect to soon launch an international phase IIb clinical trial for the treatment of Crohn’s disease patients refractory to currently available treatments. In parallel, we started the development of our second product candidate Col-Treg, for the treatment of autoimmune uveitis and obtained the classification of Innovative Medication Therapy by the European Medicines Agency. This program, for which we expect to start a clinical trial phase I/II in 2015 to establish the proof of concept, will not only allow TxCell to expand its portfolio of cellular immunotherapy products, but also to demonstrate effectiveness and potential of its ASTrIA development platform.”
Highlights of the first half of 2014:
- Launch of Col-Treg development, TxCell’s second product candidate for the treatment of autoimmune uveitis, a rare disease and a leading cause of blindness in the industrialized world. Like Ovasave, Col-Treg is from TxCell’s proprietary personalized cellular immunotherapy platform – ASTrIA.
- Initial Public Offering on April 11, 2014 on Euronext market, Compartment C, with a gross fundraising of EUR 16.2 million, the correlative conversion of the convertible bond loan of EUR 3.5 million, completed by an additional gross fundraising of EUR 1.5 million in May.
- Obtaining of Advanced Therapy Medicinal Product classification by the European Medicines Agency for Col-Treg.
- Issuance by the French Agence Nationale de Sécurité du Médicament (ANSM) of the certificate of compliance with Good Manufacturing Practices (GMP) for TxCell’s cell therapy production unit in Besançon (France).
- Launch and grant award of EUR 417 thousand for the collaborative project POSITVE (including EUR 250 thousand for TxCell). This project focuses on the development and implementation of procedures to automate the first step of Ovasave manufacturing process.
- Changes in the governance of the company:
- Appointment of Ms. Marie-Yvonne Landel Meunier as an independent director, at the Board of Directors of March 7, 2014. A certified Public Accountant located in Boston since 1990, Marie-Yvonne Landel Meunier has many years’ experience of accompanying French and European biotechnology companies in the United States. As such she has extensive networks in the US financial community specializing in biotechnology;
- Change of Bpifrance Investissement permanent representative at the Board of Directors with the appointment of Ms. Marie-Laure Garrigues;
- Appointment of M. Laurent Higueret, Bpifrance Participations, as Board observer, at the Board of Directors of May 22, 2014.
Financial Results for the First Half of 2014:
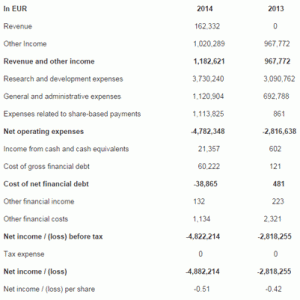
- TxCell’s revenues of EUR 162 thousand corresponds to income relating to the payment of EUR 1 million from Ferring International Center at the signature of the partnership signed on December 2013, the recognition of which is spread until December 31, 2016.
- Other operating revenues correspond to public funding for research expenditures. As of June 30, 2014, they consist of a research tax credit estimate of EUR 988 thousand for the first half of 2014 (against EUR 968 thousand as of June 30, 2013) and receivable grant revenues of EUR 31 thousand.
- The increase of operating expenses can be explained mainly by the launch of Col-Treg development, the upcoming start of the phase IIb clinical study with Ovasave and employer contributions on stock-option subscriptions granted during the first half of 2014. The recognition of expenses due to share-based compensation related to the incentive plan granted to employees and directors for EUR 1.1 million can also be noted.
- Net loss as of June 30, 2014 amounts to EUR 4,822 thousand against EUR 2,818 thousand for the period ended June 30, 2013, i.e. an increase of EUR 890 thousand excluding expenses related to share-based payments.
- As of June 30, 2014, cash and cash equivalents amounted EUR 17.4 million, against EUR 0.7 million as of December 31, 2013. This increase is mainly due to the receipt of the first milestone from the partnership with Ferring of EUR 1 million, the conversion of the convertible bond loan of EUR 3.5 million, the initial public offering of EUR 16.2 million gross, an additional gross fundraising of EUR 1.5 million and receipt of the research tax credit 2013 of EUR 1.8 million. This strong cash position provides good medium-term financial visibility for TxCell.
The income statement as of June 30, 2014, is as follow (in euros):
6 month period ended June 30
Other event:
- On September 4, 2014, TxCell has been selected by the Conseil Scientifique des Indices to be included in the CAC Small, CAC Mid & Small and CAC All-Tradable index. TxCell was also incorporated into the Next Biotech index, an index dedicated to the biotechnology sector in Europe. Integrating TxCell in these indices should increase the visibility of the company to potential new investors.
Next financial event:
Third quarter 2014 revenues on Tuesday November 4, 2014 (after market
Related conditions
Crohn’s disease