Presence of IGFBPs correlates with tumour-promoting effects of MMP9 in breast cancer
Posted: 29 May 2015 | Victoria White
A team of researchers has found that the presence of IGFBP-1 interacted with MMP9 to promote breast cancer tumour formation in mouse models…
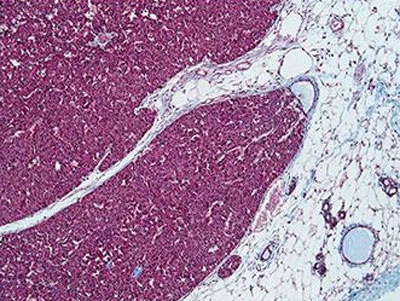

On the left, a breast cancer type called luminal; on the right, a type called triple-negative/basal. The luminal microenvironment has less fibrosis (scar tissue, coloured purple) and contains few inflammatory cells embedded within these fibrotic areas or in the surrounding fatty tissue (white). The triple-negative microenvironment contains more inflammatory cells and more fibrosis. Targeting a protein called MMP9, which promotes cancer and is linked with poor prognosis, slowed the course of the triple-negative/basal breast cancer, the one with the more profoundly altered microenvironment, but had no impact on the luminal cancer. CREDIT: Egeblad Lab, CSHL
A team of researchers led by Associate Professor Mikala Egeblad of Cold Spring Harbor Laboratory (CSHL) have found that two different mouse models of breast cancer progressed differently based on characteristics of the tumour microenvironment.
The tumour microenvironment includes cells and extracellular molecules that support the tumour’s growth. Egeblad and her team looked at two types of breast cancer driven by different mutations, and found very different microenvironments. One common factor was the presence of an extracellular protein called matrix metalloproteinase 9 (MMP9). It was expressed at similar levels in tumours from both breast cancer mouse models.
MMP9 has been linked to the progression of many cancer types
MMP9 previously has been linked to the progression of many types of cancers. When the researchers deleted the Mmp9 gene, they found that the absence of the MMP9 protein delayed tumour onset only in one mouse model, and had no effect in the other model.
Egeblad and her team found that whether MMP9 promoted cancer or not depended on the tumour microenvironment. Specifically, on the presence of another molecule that MMP9 is known to act on, called insulin-like growth factor binding protein 1 (IGFBP-1). “If IGFBP-1 is not there, MMP9 didn’t really have an effect, but if it’s there, then MMP9 has a role,” says Egeblad. This suggests that IGFBP-1 interacts with MMP9 to promote tumour formation.
MMP9 degrades IGFBPs and releases the growth factors, accelerating cancer progression
IGFBP-1 binds insulin-like growth factors (IGFs), which play a role in promoting cancer proliferation. “IGFBP-1 keeps the growth factors sequestered so they can’t act on the cancer cells and can’t make them proliferate,” Egeblad says. “But if MMP9 is present, it degrades these IGFBPs and releases the growth factors.” The release of the IGFs then accelerates cancer progression.
Egeblad and her team looked in human cancer databases to see if the interaction between MMP9 and IGFBPs predicted breast cancer prognosis in humans. “We found that IGF-binding proteins are associated with a good prognosis, but if MMP9 is also present, there’s no longer good association with survival,” Egeblad says.
The study’s results have implications for anti-cancer drugs that target MMPs, and may explain why previous clinical trials using MMP inhibitors have failed. The new study suggests that trials of MMP inhibitors could focus on patients whose tumour microenvironment contains IGFBPs.
More broadly, the research suggests that it may not be enough to see if a particular drug target is present in a certain type of cancer; researchers may also need to look for the presence of the molecules that the drug target acts upon.
The lab’s next goal is to look more generally at the differences in microenvironments in different types of cancer. “What we’re starting to learn now is that the microenvironments are different in different tumours, and that there is really a very intricate interplay between what’s driving the mutations in cancer cells and the type of microenvironment they build around themselves,” Egeblad says.
The research is published in Neoplasia.
Related topics
Oncology
Related conditions
Breast cancer
Related organisations
Cancer Research, Cold Spring Harbor Laboratory (CSHL)