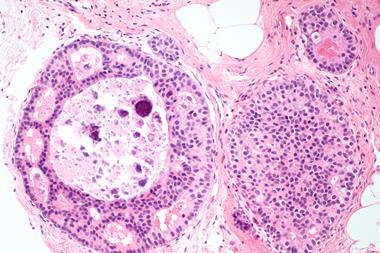
Tau drives PSP, CBD and other neurodegenerative diseases, yet there are still no disease-modifying treatments. Here, Dr Glenn Harris from the Rainwater Charitable Foundation shares how a coordinated funding effort is supporting basic research to understand tau mechanisms, improve detection and progress therapeutic development.
For decades, the amyloid-beta protein was at the heart of the neurodegenerative disease narrative. As our understanding has matured, it has become clear that another protein plays a more relevant role in causing neurotoxicity and cognitive decline – tau.1 The abnormal accumulation and aggregation of tau is the key pathological feature of Alzheimer’s disease (AD) and other neurodegenerative diseases (now broadly referred to as tauopathies).
Six major tau isoforms, each with roles in stabilising microtubules, must remain in balance, particularly the 3R and 4R isoforms, to ensure healthy neuronal function. In tau-related diseases, this balance is lost. While AD, a secondary tauopathy composed of a mix of tau isoforms, is the most prevalent and well-known tauopathy, there is a group of related, rarer diseases called primary tauopathies. Primary tauopathies involve the abnormal accumulation of tau protein as the defining pathological feature, which offers a more direct window into tau-driven pathology.2
The critical need for targeted drug discovery strategies in primary tauopathies
Examples of primary tauopathies include progressive supranuclear palsy (PSP), corticobasal degeneration (CBD), Pick’s disease (PiD) and some forms of frontotemporal dementias (FTD). For patients living with these diseases, the diagnostic journey is often long and uncertain, and the therapeutic landscape is sparse. Currently, there are no regulatory approved disease-modifying treatments targeting tau pathology,3 representing one of the most urgent unmet medical needs in an ageing society.
Primary tauopathies often exhibit unique biochemical tau isoform compositions (eg, 4R-tau in PSP and CBD or 3R-tau in PiD) and distinct anatomical distributions of pathology. This difference in tau biology highlights the need for distinct drug discovery strategies, targeting specific tau isoforms for primary tauopathies, versus strategies that may need to address the mixed protein pathology for AD.
Early detection is key to a cure
A challenge in tauopathies is the profound disconnect between the onset of pathology and the appearance of clinical symptoms. By the time a patient presents with the characteristic motor or cognitive impairments of a tauopathy, the brain has already suffered irreversible damage, given the current lack of treatments. Unlike the comparatively slow progression of AD, primary tauopathies are often cruelly aggressive, progressing from diagnosis to severe disability or even death within a few years. An effective therapeutic strategy should therefore be twofold: it must target the underlying pathology and be deployed at the earliest possible stage. Advancing early detection is not a parallel goal to finding a cure; it is the prerequisite. Our hope is to not only stop the progression of the disease, but also to potentially restore neuronal function.
The Tauopathy Challenge Workshop: seeding the next wave of discovery
To break this impasse and help discover the underlying mechanisms of tau pathology, the Rainwater Charitable Foundation (RCF) – alongside our partners the Alzheimer’s Association, the Aging Mind Foundation and CurePSP – have embarked on a new approach. We believe that to solve the most intractable problems, we must move beyond siloed efforts and instead pool our resources, expertise and networks. The Tauopathy Challenge Workshop is the latest milestone in this strategic, collaborative endeavour to build a robust therapeutic pipeline and deliver hope to patients.
The historical lack of progress in treating primary tauopathies can be largely attributed to a cycle of market failure. These neglected and underfunded neurodegenerative diseases have been perceived by many as too rare and difficult to study. As a result, foundational high-risk, high-reward research projects focused on target identification, validation and tool development have languished. This is precisely where strategic philanthropy – and specifically a partnership model – has been transformative.
The Tauopathy Challenge Workshop represents the culmination of this partnership model in action. It is not a typical academic conference but an active, small assembly of selective investigators who do not shy away from sharing unpublished data, challenging assumptions and building new collaborations.
The Tauopathy Challenge Workshop represents the culmination of this partnership model in action. It is not a typical academic conference but an active, small assembly of selective investigators who do not shy away from sharing unpublished data, challenging assumptions and building new collaborations. The research projects supported by this challenge are designed to be transformative. While representing a diverse range of scientific approaches, the funded projects address the field’s most pressing challenges.
Starting with the inaugural convening in 2023, the Workshop has focused on the translational and mechanistic gaps currently impeding progress in the field of neurodegeneration. A key example is the emerging paradigm in tau pathology: tau is not a monolith. The protein misfolds into distinct conformations, or strains, which are believed to dictate the specific cell type affected and the resulting clinical presentation. At the 2025 Workshop, investigators focused heavily on researching the molecular and cellular basis of these pathological strains, seeking to understand why, despite sharing the same isoform, one tau strain leads to PSP while another leads to CBD. This work is essential for developing strain-specific targets for biomarker or therapeutic development.
In addition to strain research, several projects are exploring the mechanisms of tau propagation between cells, the role of specific post-translational modifications in tau toxicity, the complex interplay between misfolded tau and the brain’s immune system, and the role of copathologies present in various stages of a patient’s progression.
Ultimately, by convening these leading experts, the Workshop forged a unique ecosystem of open science and collaboration, significantly accelerating the pace of discovery for the entire field. The RCF’s 2025 Tauopathy Challenge Workshop dedicated $2.5 million to five basic science programmes. This cohort of grants represents a strategic evolution in neurodegeneration research, recognising that enhancing our understanding of tau’s interactome – the web of proteins and cells it partners with – is the clearest path to developing the next generation of effective therapeutics.
A major theme across the awards is addressing the indispensable role of copathology: a recognition that tau rarely acts in isolation. Dr Eleanor Drummond (University of Sydney) will tackle this directly, aiming to uncover how abnormal protein aggregation beyond tau contributes to PSP, CBD and PiD. Using advanced protein enrichment and mass spectrometry approaches, her research will map other proteins prone to pathological clumping, determining if tau is the direct driver of their misfolding or if they represent independent disease mechanisms. This work is crucial because every newly identified protein or mechanism provides a potential fresh, unexploited biomarker or drug target.
Also studying copathology is Dr Wilfried Rossoll (Mayo Clinic), who is investigating how transactive response DNA binding protein 43 kDa (TDP-43), another well-known amyloid-forming protein, specifically influences tau pathology in PSP and CBD. Utilising spatial proteomics in human tissue alongside mechanistic studies in mouse brain tissue models, Dr Rossoll is working to map the molecular dialogue between TDP-43 and tau. Given that TDP-43 may accelerate or alter tau’s toxicity, understanding this interaction is vital for therapeutic development. Any successful drug must either account for or directly target this interplay to meaningfully slow disease progression.
The following two grants investigate the mechanisms of pathological protein interaction and seeding. Dr Leonard Petrucelli (Mayo Clinic) and Dr Michael Ward (National Institutes of Health) are collaborating to explore the role of transmembrane protein 106B (TMEM106B) fibrils. TMEM106B is a protein associated with lysosomal function and Dr Petrucelli’s team hypothesises that its fibrils actively promote tau seeding and potentially worsen PSP symptoms. This research aims to understand the propagation of the disease by identifying a possible accelerator of tau pathology. If TMEM106B is confirmed as an instigator of the pathological spread of toxic tau tangles, an antibody or small molecule capable of neutralising it could function as a potent therapeutic brake on the disease’s progression.
The resulting discoveries promise not only better diagnostic biomarkers – especially for diseases currently difficult to distinguish – but also the diverse therapeutic candidates urgently needed to treat these disorders.
Building on this structural idea is Dr Vijay Rangachari (The University of Southern Mississippi), who is investigating the possibility of hybrid amyloids formed by the co-aggregation of TDP-43 and tau. While these proteins are known to form toxic clumps independently, Dr Rangachari’s unique hypothesis suggests they can fuse into new, hybrid structures with distinct and potentially more potent pathological characteristics. Identifying the unique molecular structure of these deposits in human tissue is the first step towards developing highly specific diagnostic antibodies. A therapeutic strategy that targets a hybrid aggregate might neutralise two major pathologies with a single intervention.
Finally, Dr Mikael Simons (German Center for Neurodegenerative Diseases, DZNE) pivots focus from the internal mechanics of the neuron to the extracellular environment and supporting cast of the brain, specifically glial cells and lipid transport. Dr Simons is investigating how non-neuronal cells, like oligodendrocytes, and factors such as myelin damage and lipoproteins like apolipoprotein E (APOE) contribute to tau aggregation and propagation. Glial cells are often responsible for inflammation and clearance of cellular debris and misfolded proteins. If disruptions in myelin or lipid transport systems are causing the glial cells to malfunction, this provides a completely different therapeutic entry point. Instead of trying to fix a faulty neuron, a treatment could focus on restoring the health and function of the brain’s support system, making the environment inhospitable to tau pathology.
In summary, the 2025 Tauopathy Challenge Workshop grantees are collectively steering the field towards a multi-pronged therapeutic strategy and a more actionable understanding of neurodegeneration. This basic science investment moves decisively beyond the single-target paradigm by prioritising complex areas such as copathology, hybrid mechanisms and the non-neuronal context. The resulting discoveries promise not only better diagnostic biomarkers – especially for diseases currently difficult to distinguish – but also the diverse therapeutic candidates urgently needed to treat these disorders.
The RCF’s strategic view: a pathfinder for all neurodegeneration
The RCF’s focus on primary tauopathies is a deliberate and strategic component of its broader mission to end all neurodegenerative diseases. We view the primary tauopathies as pathfinder diseases. Because their pathology is almost exclusively driven by tau – unlike the complex, mixed-pathology of late-onset AD – primary tauopathies provide a focused model to study tau-driven neurodegeneration, which may make them valuable for testing new therapeutic hypotheses.
The mechanisms, models and tools validated in PSP or CBD will not remain confined to those diseases. By funding the foundational science of tau biology, our collaboration is enabling the development of emerging therapeutic strategies, from small molecules and antisense oligonucleotides to immunotherapies and gene therapies. A successful anti-tau therapy, a validated blood-based biomarker, or a novel therapeutic approach developed for a primary tauopathy can be rapidly adapted and scaled for much larger patient populations of other tauopathies.
The obstacle posed by tauopathies is far too complex and urgent for any single organisation to tackle alone. The era of siloed, competitive research is failing patients. By strategically aligning our resources, we are creating a research ecosystem that is more agile, efficient and resilient. We are funding the critical science that others will not, building the tools the field desperately needs and fostering the next generation of tau researchers.
References
- Goedert M, Crowther RA, Scheres SHW, Spillantini MG. (2024). Tau and neurodegeneration. Cytoskeleton, 81(1), 95–102. https://doi.org/10.1002/cm.21812
- Chung De.C, Roemer S, Petrucelli L, et al.Cellular and pathological heterogeneity of primary tauopathies. Mol Neurodegeneration 16, 57 (2021). https://doi.org/10.1186/s13024-021-00476-x
- Harris GA, Hirschfeld LR, Gonzalez MI, et al.Revisiting the therapeutic landscape of tauopathies: assessing the current pipeline and clinical trials. Alz Res Therapy 17, 129 (2025). https://doi.org/10.1186/s13195-025-01775-x
Meet the author
 Glenn A Harris, PhD, is an established contributor across the drug development ecosystem for the Rainwater Charitable Foundation, driving efforts to advance promising targets from discovery to the clinical trial phase. He is notably involved in initiatives related to neurodegenerative disorders, where he emphasises the critical need for well-defined targets, drugs and biomarkers to accelerate therapeutic breakthroughs. Dr Harris has authored numerous publications detailing the therapeutic landscape and novel methods for molecular analysis in drug and biomarker development.
Glenn A Harris, PhD, is an established contributor across the drug development ecosystem for the Rainwater Charitable Foundation, driving efforts to advance promising targets from discovery to the clinical trial phase. He is notably involved in initiatives related to neurodegenerative disorders, where he emphasises the critical need for well-defined targets, drugs and biomarkers to accelerate therapeutic breakthroughs. Dr Harris has authored numerous publications detailing the therapeutic landscape and novel methods for molecular analysis in drug and biomarker development.
Topics
- Assays
- Biomarkers
- Companies
- Disease Research
- Dr Glenn A Harris (Drug Discovery Lead at Rainwater Charitable Foundation)
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Targets
- Genomics & Sequencing
- High-Throughput Screening (HTS)
- Imaging & Diagnostics
- Informatics
- Microscopy
- Molecular Biology
- Neurological disorders
- Neuroprotection
- Neurosciences
- Pathology & Molecular Medicine
- Protein Expression
- Proteomics
- Rainwater Charitable Foundation
- Tools and techniques
- Translational Science