Role of let-7 in neuroblastoma offers a new angle on treatment
Posted: 8 July 2016 | Victoria White, Digital Content Producer | No comments yet
A study has found that let-7 plays a central role in curbing neuroblastoma and could focus efforts to find a targeted, nontoxic alternative to chemotherapy…
A new study has found that a microRNA, called let-7, plays a central role in curbing neuroblastoma and could focus efforts to find a targeted, nontoxic alternative to chemotherapy.
The study unifies several theories about neuroblastoma and has implications for other solid tumours in which let-7 is lost.
The let-7 family of microRNAs is known to be involved in both stem-cell differentiation and tumour suppression. Recent research had implicated LIN28B, a protein that inhibits let-7 maturation, in neuroblastoma. But the new study found that LIN28B is only one of several cancer mechanisms that involve let-7 suppression.
“We’re showing that let-7 inhibition is central to the development of this disease,” explained John Powers, Boston Children’s Hospital. “So critical in fact that neuroblastoma uses at least three distinct ways of eliminating it.”
Powers and colleagues first demonstrated that genetic amplification of the oncogene MYCN allows neuroblastomas to sequester let-7, taking it out of circulation.
“Children with a MYCN-amplification event, which occurs in about 25 percent of cases, have the poorest prognosis,” said Powers. “A big question in neuroblastoma research has been, ‘why do you need so much MYCN mRNA?’ We found that MYCN is expressed at such high levels that it is able to sponge up otherwise functional let-7.”
Genetic loss of let-7 tracks with poor outcomes
The researchers also showed that chromosome deletions associated with neuroblastoma (in the 11q and 3p regions) are home to multiple let-7 family members and that genetic loss of let-7 tracks with poor outcomes.
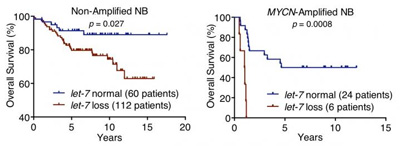
These survival curves show that chromosomal let-7 loss reduces survival in both MYCN-amplified and non-MYCN-amplified neuroblastoma. With a larger patient sample, curves such as this could help predict patient survival based on these genetic parameters. CREDIT: John Powers, PhD
“Genetic loss of let-7 was not widely appreciated in neuroblastoma,” said Powers. “People knew about chromosomal losses and were looking for a tumour suppressor, but they were mainly looking at the protein-coding genes, not microRNAs.”
Interestingly, data from 202 patients with neuroblastoma showed that chromosomal loss of let-7 rarely occurred in patients with MYCN amplification. The two cancer-promoting events, both suppressing let-7, were nearly mutually exclusive, meaning most neuroblastomas involved either one or the other.
“From the cancer’s point of view, you have to deal with let-7,” Powers explained. “If MYCN is not amplified, the tumour instead just loses it genetically. In either case, let-7 is mitigated.”
Likewise, if let-7 is eliminated through chromosomal loss, MYCN amplification becomes unnecessary for the cancer to spread.
Strategies focused on restoring let-7 could provide a fresh, low-toxicity approach to neuroblastoma. Let-7 itself could potentially be a drug, if the challenges of delivery can be overcome.
Related organisations
Dana-Farber Cancer Institute


