Scientists have discovered that adding a simple cocktail of three common amino acids to lipid nanoparticle injections can dramatically improve the delivery of mRNA and CRISPR therapies.
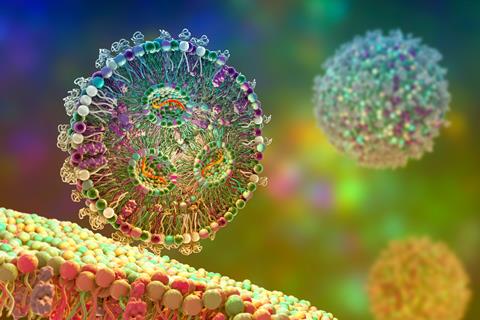
Lipid nanoparticles (LNPs) are best known as the delivery system used in COVID-19 mRNA vaccines, but they could soon power a much broader range of medical treatments. Scientists are exploring how these tiny particles can deliver therapeutic mRNA into cells to treat diseases, as well as transport CRISPR gene-editing tools that can correct harmful genetic mutations.
Yet a persistent obstacle has slowed progress. For LNP-based therapies to work, the nanoparticles must fuse with cell membranes and release their cargo inside cells. This step works efficiently in laboratory experiments but is far less effective inside the human body.
But now, researchers from Biohub believe they have identified a surprisingly straightforward way to overcome this.
Amino acids boost delivery
A group of researchers led by Daniel Zongjie Wang and Shana Kelley found that adding three common amino acids alongside LNP injections can dramatically improve delivery performance.
The team discovered that a mixture of methionine, arginine and serine increased mRNA delivery by up to 20-fold. It also boosted CRISPR gene-editing efficiency from around 25 percent to nearly 90 percent after a single dose.
The team discovered that a mixture of methionine, arginine and serine increased mRNA delivery by up to 20-fold.
"Gene editing and mRNA-based therapies will play increasing roles in the medicine of the future but they require LNPs to reach and enter cells,” said Kelley, President of Bioengineering at Biohub and Head of Biohub Chicago. “Any LNP formulation being developed today could potentially benefit from our approach.”
The research grew from a broader effort by Kelley’s team to study biological processes in laboratory conditions that more closely resemble the human body.
“That’s exactly what led us here,” said Wang, who leads Biohub’s Spatiotemporal Omics Group. “By asking why LNPs perform so differently in the physiological milieu of the body, we found a surprisingly simple answer that could make a wide range of mRNA and gene editing therapies substantially more effective.”
A metabolic bottleneck
Most previous research has focused on improving the nanoparticles themselves. Scientists have screened hundreds of new lipid molecules and used artificial intelligence to explore billions of potential combinations in search of better delivery systems. Despite these efforts, clinical delivery efficiency has remained relatively modest.
Scientists have screened hundreds of new lipid molecules and used artificial intelligence to explore billions of potential combinations in search of better delivery systems.
The Biohub researchers instead examined whether the cells receiving the nanoparticles might be the limiting factor.
“The field has spent enormous effort engineering nanoparticles,” said Wang. “We found, however, that the cell’s own metabolic state is an equally important – and addressable – part of the equation.”
The team discovered that the metabolic environment plays a central role. Standard laboratory cell culture media contain nutrient levels far higher than those found in human blood, which helps explain why LNPs perform well in laboratory settings.
When the researchers grew cells in a medium designed to mimic the composition of human plasma, nanoparticle uptake dropped by between 50 and 80 percent.
Further metabolic and genetic analysis demonstrated that amino-acid-related pathways were significantly reduced under these more realistic conditions, suggesting that cells in the body operate on a leaner metabolic profile that limits their ability to absorb nanoparticles.
Dramatic results in animal tests
To counteract this effect, the researchers screened different nutrient combinations and developed a supplement containing methionine, arginine and serine.
When administered together with LNPs, the cocktail produced a five-to-twenty-fold increase in target protein expression across multiple cell types in laboratory experiments and animal models. The improvement appeared across three different delivery routes: intramuscular, intratracheal and intravenous injection.
Further analysis showed the supplement enhances a cellular uptake pathway that effectively widens the gateway through which nanoparticles enter cells.
Further analysis showed the supplement enhances a cellular uptake pathway that effectively widens the gateway through which nanoparticles enter cells.
The team tested the approach in a mouse model of acetaminophen-induced acute liver failure, a condition that is also the leading cause of drug-related liver failure in humans. Mice treated with LNP-delivered growth hormone mRNA alone had a survival rate of just 33 percent. When the amino acid supplement was added, all of the mice survived.
In a separate experiment targeting lung tissue, CRISPR-Cas9 components delivered via LNPs achieved gene-editing rates of 20 to 30 percent without the supplement. With the amino acid mixture, editing efficiency jumped to between 85 and 90 percent after a single treatment, a level that researchers say could be transformative for diseases such as cystic fibrosis.
A simple path to clinical use
One reason the discovery could move quickly toward clinical application is its simplicity. The supplement uses pharmaceutical-grade amino acids that are already produced at industrial scale and widely considered safe.
Rather than redesigning nanoparticles or genetically altering target cells, researchers say the mixture could simply be added to existing LNP injections, potentially improving the effectiveness of a wide range of emerging mRNA and gene-editing therapies.




















