A new study proves that organoid drug testing is highly accurate at predicting effective treatments and finds a novel therapeutic option.
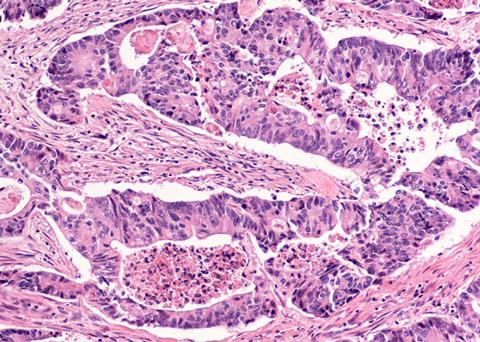
A world-first study led by scientists from the Walter and Eliza Hall Institute of Medical Research (WEHI) has proved that organoid drug testing can predict how patients with advanced bowel cancer will respond to treatment with 90 percent accuracy.
Bowel cancer, also named colorectal cancer, is the second leading cause of cancer-related deaths worldwide. Although bowel cancer cases can be treated successfully if detected early, a lack of symptoms means that less than half of patients are diagnosed at the initial stages. The number of treatment options available are increasing but there is a limited ability to predict which therapy will be most effective for individual patients.
Dr Peter Gibbs, co-lead researcher, medical oncologist and a WEHI Laboratory Head, stated that the discovery could stop the current trial-and-error process that selects a cancer treatment for patients. “Each time you give a patient an ineffective treatment, you lose two to three months on something that won’t work.”
He further explained: “Many patients with advanced bowel cancer only get one or two chances at treatment. Knowing what is most likely to work before they start treatment would make a significant difference to their survival outcomes and quality of life.”
The tumour organoids of 30 patients with advanced stages of bowel cancer were grown. Tumour organoids mimic the characteristics of the cancer from which they were created, including sensitivity to drug treatment. This enabled the researchers to pre-test chemotherapy drugs in a clinical feasibility trial. Corresponding author Dr Oliver Sieber, Associate Professor and a WEHI Laboratory Head, commented: “Our study showed organoid drug testing was able to predict treatment responses for study patients with a notable 83 percent accuracy…Importantly, pre-testing showed the therapies that won’t work with over 90 percent accuracy.”
Furthermore, the scientists used the organoids to test the effectiveness of chemotherapy drugs that are not typically used for bowel cancer patients and discovered that two patient organoids were sensitive to a drug that is often used for breast and bladder cancers, which could be a potential new therapeutic option.
Currently, a clinical trial is being developed based on the results, which will be the first to validate organoid drug testing as an accurate way to aid treatment selection for bowel cancer patients.
This study was published in Cell Reports Medicine.