Northwestern University scientists have discovered a hidden ‘control switch’ in the TRPM5 protein, which helps regulate taste, blood sugar and gut health and could lead to new therapies for targeting diabetes and obesity.
Northwestern University researchers have identified a previously hidden molecular ‘control switch’ inside a protein that plays a crucial role in sensing taste, regulating blood sugar and defending the gut.
The study demonstrates that the protein TRPM5, long thought to be activated solely by increases in calcium inside cells, can in fact be directly controlled by small molecules. This discovery could lead to potential new therapies for metabolic and digestive disorders.
A dual-use control system
TRPM5 acts as a signal amplifier in many cell types. When open, it allows sodium ions to flow through, enabling cells to send electrical signals that regulate essential biological processes. On the tongue, TRPM5 helps detect sweet, bitter and umami flavours; in the pancreas, it supports insulin release after meals; and in the gut, it helps sense nutrients and control immune defence.
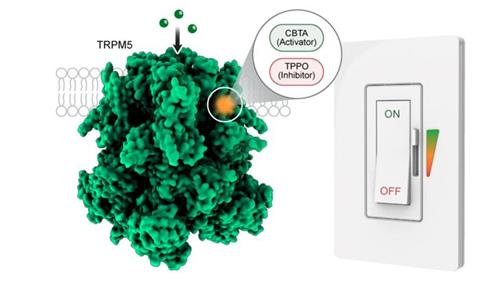
The scientists identified a ‘hidden pocket’ within TRPM5 that functions like a universal remote control.
The scientists identified a ‘hidden pocket’ within TRPM5 that functions like a universal remote control. One molecule, called CBTA, fits into the pocket and opens the protein – analogous to pressing a ‘power’ button. Another molecule, TPPO, binds to the same pocket but shuts the channel down, like pressing ‘mute.’
“The molecules look alike and bind to the same pocket but play opposite roles,” said Juan Du, co-leader of the study. “This pocket has never been reported before.”
When TRPM5 is activated by a molecule, it becomes extra sensitive to calcium, responding to tiny changes that would not normally affect it. This means the pocket can not only control the protein but also enhance its activity.
 This illustration shows how the TRPM5 channel acts as a dual purpose control site. Credit: Juan Du/Wei Lu/Northwestern University.[/caption]
This illustration shows how the TRPM5 channel acts as a dual purpose control site. Credit: Juan Du/Wei Lu/Northwestern University.[/caption] Unlocking therapeutic potential
Because TRPM5 links taste, metabolism and gut health, the ability to fine-tune its activity could have far-reaching medical applications. Potential therapies include boosting insulin release to improve glucose control in diabetes, modulating taste perception to curb food cravings and regulating gut immune signalling to reduce inflammation.
Because TRPM5 links taste, metabolism and gut health, the ability to fine-tune its activity could have far-reaching medical applications.
“TRPM5 is involved in metabolic disorders, including type 2 diabetes and obesity,” said Wei Lü, who co-led the study with Du. “If we can identify drugs that activate this channel, then we could promote insulin production to treat diseases that have problems with insulin secretion. Now that we have an overall architecture of what TRPM5 looks like and know how to activate and inhibit it, we have provided a basis for future drug development."
A foundation for future research
The research builds on a 2021 study by Lü and Du which provided the first high-resolution images of TRPM5 and identified potential drug targets.
For the current study, the team used cryo-electron microscopy (cryo-EM) and electrophysiology to visualise TRPM5’s inner workings at near-atomic resolution. The experiments revealed how molecules can act as either accelerators or brakes, highlighting a previously unrecognised level of molecular control.
The findings provide not only fundamental insights into how the body senses taste and manages metabolism but also a concrete framework for developing future therapies. By precisely targeting TRPM5, scientists could create drugs capable of controlling insulin release and gut immunity – potentially offering new treatments for diabetes, obesity and inflammatory gut conditions.
Topics
- Analytical Techniques
- Drug Delivery
- Drug Development
- Drug Discovery
- Drug Discovery Processes
- Drug Targets
- Drugs and therapies
- Inflammatory Gut Conditions
- Ion Channels
- Juan Du (Professor at Northwestern University)
- Metabolic disorders
- Molecular Biology
- Molecular Targets
- Neurological disorders
- Northwestern University
- Pharmacology
- Protein Expression
- Small Molecules
- Structural Biology
- Translational Science