Artificial chemical receptor developed to assist in T cell engineering
Posted: 15 April 2019 | Drug Target Review | No comments yet
Engineered T cell immunotherapy (such as chimeric antigen receptor T cell (CAR-T) and T cell receptor T cell (TCR-T) therapy), could potentially be used as a therapeutic strategy for tumour treatment.
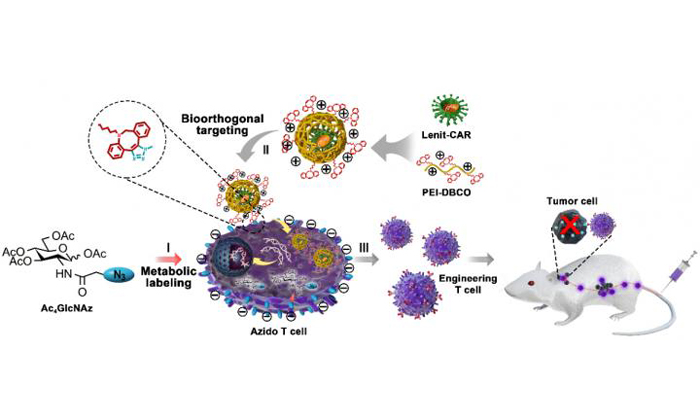

Application of artificial chemical receptor in CAR-engineered T cell manufacturing and immunotherapy of tumour (credit: CAI Lintao)
Led by Professor Cai Lintao at the Shenzhen Institutes of Advanced Technology (SIAT) of the Chinese Academy of Sciences, and other collaborators, a research team has developed a “safe, efficient and universal” strategy based on bioorthogonal chemistry and glycol-metabolic labeling for viral-mediated engineered T cell manufacturing
However, the genetic manipulation of primary T cells remains inefficient, especially during the clinical manufacturing process, and there’s an urgent need to develop a reliable method for the preparation of engineered T cells.
“We found that this artificial chemical receptor effectively facilitated viral binding to T cells and elevated the transduction efficiency of the lentivirus from 20 to 80 percent without any effect on T cell proliferation and activity,” Professor Lintao explained. “This artificial chemical modification was also appropriate for introducing other heterologous genes into T cells, including GPF, CAR and TCR, indicating a great potency for universal T cell engineering.”
The technique has also been demonstrated to be safe for human primary T cells without interference from cell expansion or antitumor functions.
When put into the CAR-T preparation, the PEI-DBCO/azide-glucose system increased the yield of CAR T cells and boosted their antitumor effect in both the in vitro and the B lymphoma xenograft mouse model with a low dose of CAR-T cells, thus reducing clinical adverse effects.
“This artificial chemical labeling strategy is an effective, safe and easy upgrade for viral-based gene manipulation of human primary T cells, thereby showing great potential for clinical engineered T lymphocyte manufacturing, including CAR-T and TCR-T cell therapy,” Professor Lintao concluded.
The research team’s findings were published in Advanced Functional Materials.
Related topics
Disease Research, Gene Therapy, Immunotherapy
Related conditions
Tumours
Related organisations
Chinese Academy of Sciences
Related people
Professor Cai Lintao