Nutlin-3 reverses damage from the mutation that causes fragile X
Posted: 27 April 2016 | Victoria White, Digital Content Producer | No comments yet
Fragile x mice models treated with Nutlin-3 regained the ability to remember what they had seen, and smelled, in their first visit to a test chamber…
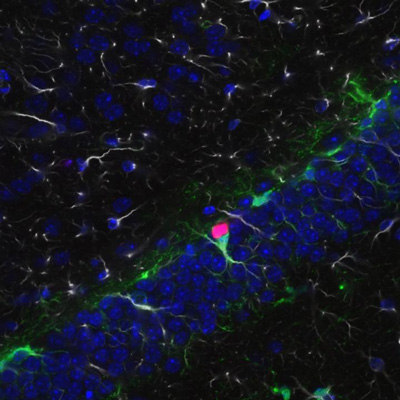
A University of Wisconsin-Madison researcher who studies fragile X has used an experimental drug to reverse – in mice – damage from the mutation that causes the syndrome.
Fragile X affects one boy in 4,000 and one girl in 7,000. It is caused by a mutation in a gene that fails to make the protein FMRP. In 2011, Xinyu Zhao, a professor of neuroscience, showed that deleting the gene that makes FMRP in a region of the brain that is essential to memory formation caused memory deficits in mice that mirror human fragile X.
The deletions specifically affected neural stem cells and the new neurons that they form in the hippocampus.
Tantalizingly, Zhao’s 2011 study showed that reactivating production of FMRP in new neurons could restore the formation of new memories in the mice. But what remained unclear was exactly how the absence of FMRP was blocking neuron formation, and whether there was any practical way to avert the resulting disability.
Now, in a new study, Zhao and her colleagues at the Waisman Center at UW-Madison have detailed steps in the complex chain reaction that starts with the loss of FMRP and ends up with mice that cannot remember what they had recently been sniffing.
Nutlin-3
This study’s newfound understanding of the biochemical chain of events became the basis for identifying an experimental cancer drug called Nutlin-3, which blocks the reaction.
In the new study, mice with the FMRP deletion took Nutlin-3 for two weeks. When tested four weeks later, they regained the ability to remember what they had seen, and smelled, in their first visit to a test chamber. Statistically, the memory capacities of normal mice and fragile X models that were treated with Nutlin-3 were identical.
Nutlin-3, which can block the last stage of the chain reaction set off by a mutation in the FMRP gene, is in Phase 1 trial for the treatment of the eye cancer retinoblastoma. Finding a new use for a drug that is approved, or that like Nutlin-3 and several derivatives, has entered the approval process, may shorten the lengthy FDA process, says Zhao.
The dose used in the trial – only 10 percent of the dose proposed for cancer chemotherapy –caused no apparent harm, she says. “We measured body weight and activity. So far, the mice look healthy and happy.”
Because more than one-third of fragile X patients are also diagnosed with autism, the study may shed light on that condition.
In any case, it’s far too soon to declare victory over fragile X, Zhao stresses. “There are many hurdles. Among the many questions that need to be answered is how often the treatment would be needed. Still, we’ve drawn back the curtain on fragile X a bit, and that makes me optimistic.”
Related topics
Stem Cells
Related organisations
University of Wisconsin-Madison






