Study defines small-cell lung cancer subtypes and therapeutic vulnerabilities
Posted: 22 January 2021 | Victoria Rees (Drug Target Review) | No comments yet
Researchers have identified four unique subtypes of small-cell lung cancer (SCLC) following the characterisation of cell types.
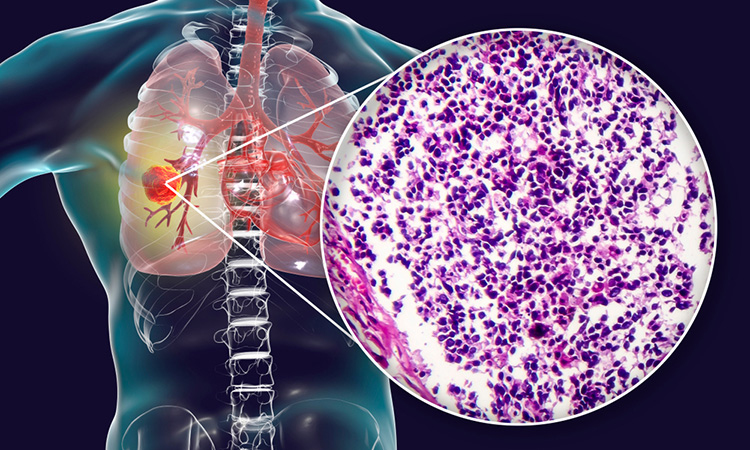

Researchers have developed the first comprehensive framework to classify small-cell lung cancer (SCLC) into four unique subtypes based on gene expression and have identified potential therapeutic targets for each type. The study was conducted at The University of Texas MD Anderson Cancer Center, US.
“For decades, SCLC has been treated as a single disease because the tumours all look similar under the microscope, even though they behave very differently,” said Associate Professor Lauren Averett Byers, senior author of the study. “Our study provides a transformative new system to define four major groups of SCLC and for the first time, an avenue for personalised treatment of the second most common type of lung cancer.”
Although previous research has identified three possible subtypes of the cancer based on transcription factors, which indicate whether particular genes are turned “on” or “off,” a large number of SCLC tumours did not fit into one of the three groups. Rather than trying to apply a hypothesis to the remaining tumours, the team took an unbiased bioinformatics approach. They applied non-negative matrix factorisation which led to a 1,300 gene “signature” that confirmed the three previously observed groups (A, N and P), plus a previously unrecognised fourth group (I) with a unique immune landscape.
The first three groups are defined by activation of the ASCL1 (SCLC-A), NEUROD1 (SCLC-N) and POU2F3 (SCLC-P) genes. The fourth type, SCLC-I, is characterised by an inflamed gene signature with a high expression of multiple immune genes, including significantly greater levels of genes indicating the presence of CD8-positive cytotoxic T cells.
One of the known challenges of this cancer is that it often develops resistance to treatment, even after an initial response. To determine if “subtype switching” causes resistance, the authors used single-cell RNA sequencing to evaluate tumour evolution in a series of patient-derived SCLC models. The study suggests that SCLC-A tends to switch to SCLC-I after being treated with chemotherapy, which could contribute to treatment resistance.
“Our paper shows that the inflamed group has a distinct biology and environment and tends to be more responsive to immunotherapy,” Byers said. “Identifying the inflamed group is very important because so far there have not been any validated biomarkers for SCLC that predict which patients get the most benefit from immunotherapy.”
Based on recent clinical trials, immunotherapy has become part of the standard-of-care for SCLC. However, all clinical trials for the condition, including those using immune checkpoint inhibitors, have had limited success. This study could help explain why, as the results suggest different classes of drugs may be more effective in specific subtypes. For example, in the samples from this study, SCLC-I was most sensitive to immune checkpoint blockade, SCLC-A to BCL2 inhibitors, SCLC-N to Aurora kinase inhibitors and SCLC-P to PARP inhibitors.
“Immunotherapy plus chemotherapy is currently the backbone of treatment for all advanced SCLC patients, but not all patients experience the same benefit,” said Assistant Professor Carl Gay, lead author of the study. “Our results provide an opportunity to think about immunotherapy approaches that are specific to the inflamed group, which has a very different microenvironment, separately from combination approaches that might activate the immune response in the other three groups.”
The study was published in Cancer Cell.
Related topics
Oncology
Related conditions
Small cell lung cancer (SCLC)
Related organisations
The University of Texas MD Anderson Cancer Center