Scientists target protein in skeletal muscle composition
Posted: 18 September 2023 | Taylor Mixides (Drug Target Review) | No comments yet
A group of researchers from Tokyo Metropolitan University have illuminated a crucial piece of the intricate puzzle surrounding the regulation of muscle mass and metabolic processes within skeletal muscle.
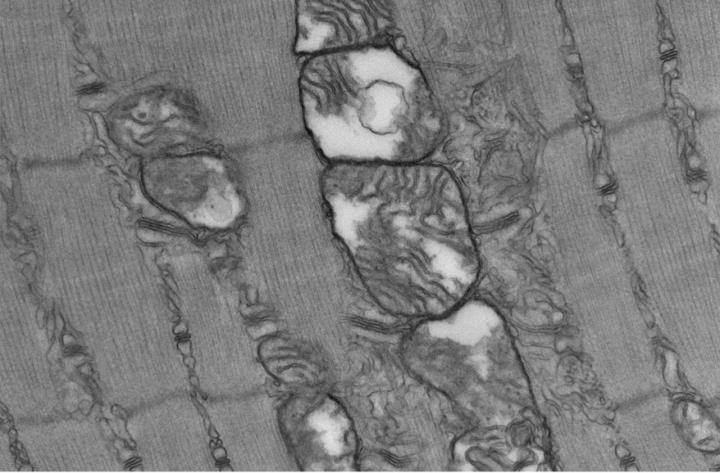
Sepsis triggers profound long-lasting ultrastructural defects in skeletal muscle mitochondria (credit: Owen et al. (CC BY 4.0)).
Their investigation, centred on Musashi-2 (Msi2), a protein that appears to hold a pivotal role in this realm, revealed profound insights into the maintenance and modulation of muscle tissue. In particular, their focus turned to the impact of Msi2 depletion, achieved through gene knockout techniques, shedding light on the profound consequences that ensue in the absence of this protein. Published here.
Skeletal muscle fibres are a marvel of adaptability, capable of responding dramatically to various stimuli. Through diligent training, individuals can bolster their muscle mass, enhance strength, and cultivate endurance. Conversely, extended periods of physical inactivity or the inexorable passage of time can usher in muscle atrophy, a condition replete with detrimental consequences for mobility and overall health. Yet, despite awareness of these phenomena, the precise mechanism orchestrating the regulation of muscle mass and strength remains an enigma that has long eluded the grasp of scientists.
In pursuit of this biological conundrum, Assistant Professor Yasuro Furuichi and his team at Tokyo Metropolitan University set out on a quest to discern the enigmatic role of Musashi-2 (Msi2). Originally identified within nerve cells, Msi2 was found to be expressed within the realm of skeletal muscle tissue. Intriguingly, its expression dwindled in the face of muscular atrophy, igniting suspicions that Msi2 was intricately involved in the development of muscle fibres. Determined to unearth the truths hidden within these molecular mysteries, the researchers embarked on a meticulous journey of inquiry.
Their journey began by extracting muscle tissue from mice and subjecting it to enzymatic processes that meticulously separated muscle fibres from the surrounding neural, vascular, and adipose tissues. Through meticulous analysis, the team unequivocally established that Msi2 was indeed expressed within the muscle fibres themselves. Furthermore, their examination of fibres harvested from calf muscles uncovered a compelling revelation: Msi2 exhibited its most robust expression within type 1, or “slow,” muscle fibres—fibres renowned for their endurance yet less renowned for explosive power compared to their type 2, or “fast,” counterparts.
Undoubtedly, the discoveries emerged when the researchers explored mice with the gene encoding the Musashi-2 protein deliberately “knocked out.” These Msi2-deficient mice presented with calf muscles characterised by a substantial reduction in mass, a pallid appearance, and diminished strength. As the team peered through the lens of a microscope, the root of this muscular decline became apparent: a marked reduction in the population of type 2a fibres, a variety of “fast” fibres distinguished by their unique blend of endurance reminiscent of “slow” fibres. Additionally, the Msi2 knockout mice demonstrated a compromised ability to metabolise sugars, mirroring the metabolic dysregulation observed in diabetes. The culprits behind this metabolic impairment were fewer myoglobin and mitochondria, both indispensable to produce cellular energy. Remarkably, the researchers found that this affliction could be rectified by reintroducing Msi2 specifically into “fast” fibres, thereby restoring the levels of myoglobin and crucial protein markers associated with mitochondrial function. This observation underscored Msi2’s role in governing proteins associated with sugar metabolism while wielding influence over the composition of different fibre types within the tapestry of skeletal muscle tissue.
Notably, type 2a fibres occupy a unique position within the spectrum of muscle fibres. They exhibit a remarkable responsiveness to training, yet they are also exceptionally prone to atrophy with advancing age. These distinguishing characteristics render any insights into the regulation of type 2a fibres of paramount importance. By exploring the intricate mechanisms by which Msi2 influences muscle development and metabolic processes, this study has ushered in a profound advancement in the quest to identify therapeutic targets for combating the decline of muscle tissue. Moreover, it lays the groundwork for the development of tailored training regimens aimed at enhancing muscle strength and resilience. In the ceaseless pursuit of knowledge, the role of Musashi-2 emerges as a crucial linchpin, offering tantalising prospects for the future of muscle science and the promotion of lifelong health and vitality.
Related topics
Gene Testing, Gene Therapy
Related organisations
Tokyo Metropolitan University
Related people
Yasuro Furuichi (Tokyo Metropolitan University)






