Dissolving implant device relieves pain without drugs
Posted: 1 July 2022 | Ria Kakkad (Drug Target Review) | No comments yet
Researchers have created a new implant device has the potential to provide an alternative to opioids and other highly addictive drugs.


Scientists from the Northwestern University, US, have developed a small, soft, flexible implant that relieves pain on demand and without the use of drugs. The first-of-its-kind device could provide a much-needed alternative to opioids and other highly addictive medications. The paper, which was recently published in Science, describes the device’s design and demonstrates its efficacy in an animal model.
The biocompatible, water-soluble device works by softly wrapping around nerves to deliver precise, targeted cooling, which numbs nerves and blocks pain signals to the brain. An external pump enables the user to remotely activate the device and then increase or decrease its intensity. After the device is no longer needed, it naturally absorbs into the body — bypassing the need for surgical extraction.
The researchers believe the device has the potential to be most valuable for patients who undergo routine surgeries or even amputations that commonly require post-operative medications. Surgeons could implant the device during the procedure to help manage the patient’s post-operative pain.
“The technology reported here exploits mechanisms that have some similarities to those that cause your fingers to feel numb when cold. Our implant allows that effect to be produced in a programmable way, directly and locally to targeted nerves, even those deep within surrounding soft tissues,” explained Professor John A. Rogers, the scientist who led the devices development.
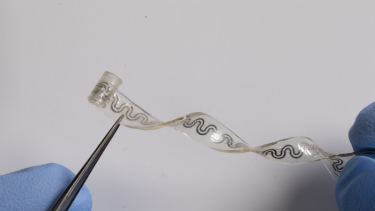

The soft flexible device bends and stretches with the body, without the need for bulky, rigid hardware
[Credit: Northwestern University].
“As you cool down a nerve, the signals that travel through the nerve become slower and slower — eventually stopping completely,” said study co-author Dr Matthew MacEwan. “We are specifically targeting peripheral nerves, which connect your brain and your spinal cord to the rest of your body. These are the nerves that communicate sensory stimuli, including pain. By delivering a cooling effect to just one or two targeted nerves, we can effectively modulate pain signals in one specific region of the body.”
To induce the cooling effect, the device contains tiny microfluidic channels. One channel contains the liquid coolant (perfluoropentane), which is already clinically approved as an ultrasound contrast agent and for pressurized inhalers. A second channel contains dry nitrogen, an inert gas. When the liquid and gas flow into a shared chamber, a reaction occurs that causes the liquid to promptly evaporate. Simultaneously, a tiny integrated sensor monitors the temperature of the nerve to ensure that it’s not getting too cold, which could cause tissue damage.
“Excessive cooling can damage the nerve and the fragile tissues around it,” Rogers said. “The duration and temperature of the cooling must therefore be controlled precisely. By monitoring the temperature at the nerve, the flow rates can be adjusted automatically to set a point that blocks pain in a reversible, safe manner. On-going work seeks to define the full set of time and temperature thresholds below which the process remains fully reversible.”
At its widest point, the tiny device is just 5 millimetres wide. One end is curled into a cuff that softly wraps around a single nerve, bypassing the need for sutures. By precisely targeting only the affected nerve, the device spares surrounding regions from unnecessary cooling, which could lead to side effects.
All components of the devices are biocompatible and naturally absorb into the body’s biofluids over the course of days or weeks, without needing surgical extraction. The bioresorbable devices are completely harmless — like absorbable stitches.
The researchers therefore conclude that at the thickness of a sheet of paper, the soft, elastic nerve cooling device is ideal for treating highly sensitive nerves.
Related topics
Technology, Therapeutics
Related conditions
Drug addiction, pain
Related organisations
Northwestern University
Related people
Professor John A. Rogers