New pathway involved in cellular protein degradation could develop LYTACs therapy and find new treatments for lysosome shortage disorders.
To target proteins involved in causing or spreading disease, typically a drug will obstruct a protein’s active site so it cannot function. However, new strategies to deal with these proteins involve sending them to different types of cellular protein degradation machinery such as a cell’s lysosomes, which act as a protein “woodchipper.”
Chemists at Stanford University discovered how one of the pathways leading to this protein “woodchipper” works furthering the understanding of new therapeutics for autoimmune diseases, age-related disorders, and treatment-resistant cancers. Also, their findings could improve therapeutics for rare but serious lysosomal storage disorders that mostly affect babies and children. Lysosomal storage diseases (LSDs) are a group of inherited metabolic disorders that are caused for the most part by enzyme deficiencies within the lysosome resulting in accumulation of undegraded substrate. This storage process leads to a broad spectrum of clinical manifestations depending on the specific substrate and site of accumulation.1
Dr Carolyn Bertozzi, the Anne T. and Robert M. Bass Professor in the School of Humanities and Sciences and Baker Family Director of Sarafan ChEM-H, said: “Understanding exactly how proteins are shuttled to lysosomes to be broken down can help us harness the innate power of a cell to get rid of proteins that cause the human body so much harm.” She continued: “The work done here is a clear look into a typically opaque intracellular process, and it’s shining a light on a new world of possible drug discovery.”
“The work done here is a clear look into a typically opaque intracellular process, and it’s shining a light on a new world of possible drug discovery.”
Steven Banik, Assistant Professor of Chemistry in the School of Humanities and Sciences, said: “The ability to understand the biology of this process means we can use inherent biology that already exists, and harness it to treat disease.” He added: “These insights offer a unique window into a new type of biology that we haven’t really understood before.”
Stopping destructive proteins
Often, proteins are helpful within the body, aiding in digestion and repairing torn muscles. However, they can also be destructive. Proteins can become part of a cancerous tumour and allow for its unrestricted growth, cause diseases like Alzheimer’s, and build up in the heart to impact how it pumps blood around the body.
The standard of therapeutic research was making drugs to stop rogue proteins, blocking their active site and therefore preventing it from interacting with a cell. Then, 20 years ago, proteolysis targeting chimeras (PROTACs) began to be utilised. PROTACS engage bad-acting proteins that are already inside a cell and send them off to be broken down in the lysosome.
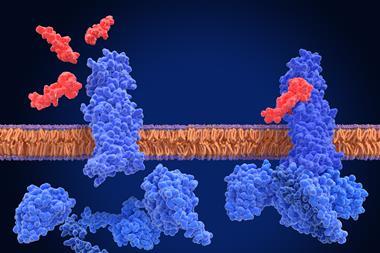
Although PROTACs are currently in clinical trials and have shown efficacy in treating cancer, they can only target a protein if it is inside the cell, which is 60 percent of the time. To reach the other 40 percent of those proteins, Stanford ChEM-H researchers pioneered lysosome targeting chimeras (LYTACs) in 2020. LYTACs find and mark proteins for destruction, that are around the cell or on a cell’s membrane.
These findings prompted a new area of research and therapeutics, but exactly how the process worked was unknown. It was also challenging to predict when LYTACs would be successful or fail to perform as expected.
Novel targets
In this study, lead author Dr Green Ahn, then a Stanford graduate student and now a postdoctoral fellow at the University of Washington Institute for Protein Design, used a genetic CRISPR screen to identify and characterise the cellular components that modulate how LYTACs degrade proteins. This screening enabled the team to find a link between the level of neddylated cullin 3 (CUL3), a protein that has a role in breaking down cellular proteins, and LYTAC efficacy. Although the precise tie is unknown, the more neddylated CUL3 present, the more effective LYTACs were.
To test which patients are more likely to respond to LYTAC therapy, the level of neddylated CUL3 could be measured. Bertozzi noted that this was a surprise finding as no previous research suggested this correlation.
The team also found proteins that block LYTACs from functioning normally. LYTACs bind to certain receptors on the outside of the cell, which they use to transfer undesirable proteins into lysosomes for degradation. However, the researchers observed that proteins bearing mannose 6-phosphates (M6Ps), sugars that decorate proteins destined for lysosomes, will seat themselves on those receptors, meaning LYTACs have nowhere to bind. By disrupting M6P biosynthesis, a higher fraction of unoccupied receptors resulted on the cell surface which could be hijacked by LYTACs.
New treatments
This study could not only aid in developing LYTACs into more effective therapeutics, but it may also lead to new and more effective treatments for lysosome shortage disorders. Enzyme replacement therapy is a frequent treatment for these disorders. This therapy uses similar pathways as LYTACs to travel to lysosomes where they can operate, so understanding how and why LYTACs work means that these enzymes could be delivered more effectively.
Bertozzi noted: “We’re learning human biology that wasn’t known before.”
This study was published in Science.
References
1 Sun A. Lysosomal storage disease overview. Annals of Translational Medicine (ATM). 2018 December [2023 November 1]; 6(24):476. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6331358/