Improved targeted ocular spectroscopy for better diagnosis
Posted: 19 December 2023 | Drug Target Review | No comments yet
In vitro and in vivo experiments highlight the advantages of a new targeted spectroscopy system for many eye diseases.
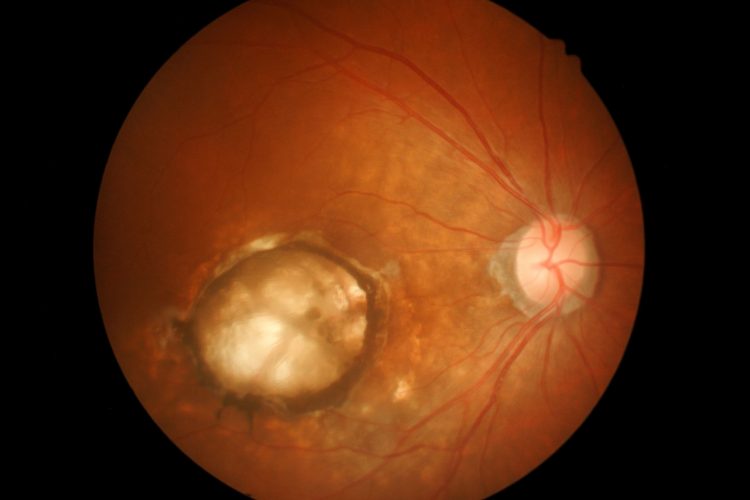
Scientists from Zilia Inc, Canada, led by Dr Dominic Sauvageau from the University of Alberta, have developed a much more flexible system for targeted spectroscopy in the back of the eye, known as the eye fundus. Numerous ocular diseases involve changes in the structure and function of the eye fundus. For example, the degeneration of ganglion cells is a defining characteristic of glaucoma. However, changes in the eye fundus are not restricted to vision-related diseases only, as some neurological conditions like Parkinson’s disease can cause changes in retinal nerves and blood flow.
Eye care professionals typically rely on colour imaging and computed tomography techniques to diagnose ocular diseases, but over the past few decades, scientists have discovered that disease-related changes in the eye fundus also modify its profiles for spectral reflectance and emittance. This means that the way in which light interacts with specific retinal structures at different frequencies can give critical diagnostic information to complement standard imaging methods.
Although various methods have been proposed, they still have significant limitations. One common issue is that most spectroscopy-based methods can only make measurements over a large region of the eye fundus, which restricts their ability to detect fine spectral changes in small retinal structures. However, techniques that make localised spectral measurements require the fixation of the patient, which can be very tedious and uncomfortable.
In the new study, the research team have developed a more flexible system for targeted spectroscopy to overcome these issues, which have key features that make it more versatile. The entire device has a series of optical elements: illumination LEDs, a colour camera, and a spectrometer, which enables three different light paths to and from the eye fundus to coexist without impeding each other.
The spectrometer section of the device focuses an LED onto a small region of the eye fundus. The position of this region can be adjusted using simple mechanical actuators to rotate the beam splitter feeding the camera and the spectrometer, making it easy to take spectral measurements from very specific anatomical structures, like the optic nerve, leakage of blood, or any type of lesion. Also, the system can be used to conduct fluorescence measurements by adjusting the illumination source, which extends its applicability to detect an even wider variety of biomarkers.
The scientists conducted both in vitro and in vivo experiments to confirm their system’s capability. The in vitro experiments involved targeting coloured areas in a reference target with a grid-like pattern and taking spectral measurements of an eye model that simulated a macula, blood vessels, a foreign body, and the optic nerve. The in vivo measurements exhibited differences in the spectral profiles of the optic nerve and the parafoveal region in the retinas of eight healthy subjects.
The study’s results highlight the many advantages of the proposed design and could pave the way to better diagnostic protocols for eye diseases. Dr Sauvageau explained: “Targeted ocular spectroscopy has the potential to assess the presence of different chromophores and fluorophores, such as haemoglobin, oxyhaemoglobin, melanin, and lipofuscin, associated with disease progression…This could open the door to changes in the way we diagnose and treat eye diseases, and targeted ocular spectroscopy could become an increasingly important tool in eye care in the coming years.”
This study was published in the Journal of Biomedical Optics (JBO).
Related topics
Biomarkers, In Vitro, In Vivo, Spectroscopy
Related conditions
Glaucoma, Parkinson's disease (PD)
Related organisations
University of Alberta, Zilia
Related people
Dr Dominic Sauvageau (University of Alberta)