Light-stimulated brain cells could aid in stroke recovery research
Posted: 1 July 2019 | Drug Target Review | 1 comment
A new ‘optochemogenetics’ research approach from scientists at the Emory University School of Medicine could aid in helping stroke victims.
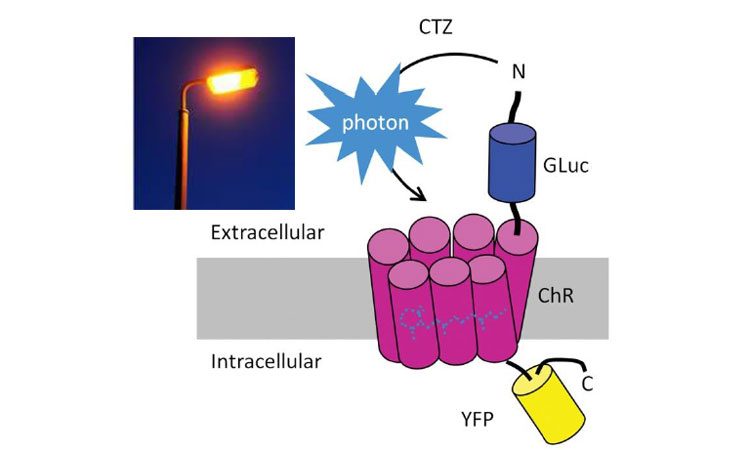

Gaussia luciferase (Gluc) is fused to the ChR protein. ChR can be activated by blue light or by light emitted by Gluc when binding to its substrate coelenterazine (CTZ). YFP = yellow fluorescent protein (credit: Shan Ping Yu).
Transplanting neural progenitor cells to restore lost functions is a promising strategy for helping stroke patients recover. However, this asks a lot of those cells. To aid in this, researchers at the Emory University School of Medicine have developed an ‘optochemogenetics’ research approach. This has shown the stimulation that transplanted cells need to flourish comes from light generated within the brain.
For the study, the researchers used a mouse model of a stroke and when the neural progenitor cells received light stimulation, it promoted functional recovery in the mice.
The researchers, led by Shan Ping Yu, MD, PhD and Ling Wei, MD wanted to figure out how to selectively stimulate brain cells non-invasively. To do this they teamed up with Jack Tung, PhD, Ken Berglund, PhD and Robert Gross, MD from the Department of Neurosurgery and the Wallace H Coulter Department of Biomedical Engineering at Georgia Tech and Emory, who had created ‘luminopsins’ (engineered proteins that are both light-sensitive and generate their own light when provided with the chemical coelenterazine).
Yu and Wei were looking for ways to coax neural progenitor cells to survive in the brain after the destruction of a stroke, working with a mouse model in which the sensory and motor regions on one side of the brain were damaged.
To do this, the scientists introduced genes encoding luminopsins into induced pluripotent stem cells, which were cultured to form neural progenitor cells. The neural progenitor cells were then delivered into the brains of mice a week after stroke.
CTZ, which emits light when acted upon by luminopsins, was then provided intra-nasally twice a day for two weeks. Intra-nasal delivery bypasses the blood-brain barrier and repeated administrations are clinically feasible, Yu points out. Bioluminescence could be detected in the cell graft area and was visible for around one hour after CTZ administration.
CTZ promoted an array of positive effects in the progenitor cells: more survival and intact axons, more connections within the brain and better responses to electrical stimulation. It also promoted recovery of function in the affected limb in the mice. In young mice, CTZ and progenitor cells together could restore use of the stroke-affected limb back to normal levels, and even in older mice, they produced partial recovery of function.
“Optogenetics is a fantastic technical tool, but it presents some barriers to clinical implementation,” said Yu. “You have the invasive fiber optic light delivery, and the limited distance of light diffusion, especially on the larger scale of the human brain.”
Delivery of cells into the brain and making them glow are complex, but they offer scientists some flexibility when designing experiments: direct light application, which can be turned on and off quickly, or the steady support of CTZ stimulation.
Luminopsins can provide “the capabilities for the cells to be activated either in a way mimicking neuronal activities of fast activation or manipulations of the channel/cell in clinical treatments,” the authors write.
The results from this study are scheduled for publication in Journal of Neuroscience.
Related topics
Cell Cultures, Protein, Regenerative Medicine, Research & Development, Stem Cells
Related conditions
Stroke
Related organisations
Emory University School of Medicine.
Related people
Jack Tung PhD, Ken Berglund PhD, Robert Gross MD, Shan Ping Yu MD PhD







awesome new trend, as a stroke victim, I hope to benefit in the future from this kind of research, which holds so much promise.