Ivermectin shows activity against COVID-19 in cell cultures
Posted: 6 April 2020 | Hannah Balfour (Drug Target Review) | 21 comments
A study has demonstrated that the anti-parasitic drug ivermectin may be effective against SARS-CoV-2, the virus which causes COVID-19.


Researchers have discovered ivermectin, an anti-parasitic drug found world-wide, can kill SARS-CoV-2, the virus causing the COVID-19 pandemic, within 48 hours.
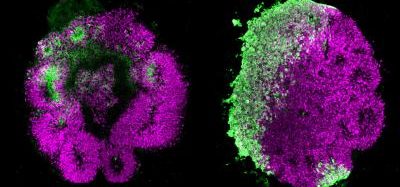
A collaborative study by Monash University’s Biomedicine Discovery Institute (BDI) and the Peter Doherty Institute of Infection and Immunity (Doherty Institute), both Australia, demonstrated that ivermectin can stop SARS-CoV-2 growth in cell cultures by eradicating all genetic information within two days. Their findings were published in Antiviral Research.
“We found that even a single dose could essentially remove all viral RNA by 48 hours and that even at 24 hours there was a really significant reduction in it,” said BDI’s Dr Kylie Wagstaff, who led the study.
Ivermectin is a US Food and Drug Administration (FDA)-approved anti-parasitic drug that has previously shown activity in vitro against a broad range of viruses, including human immunodeficiency virus (HIV), Dengue fever, influenza and Zika virus. Dr Wagstaff stated: “Ivermectin is very widely used and seen as a safe drug. We need to figure out now whether the dosage you can use it at in humans will be effective – that’s the next step.”
According to Dr Wagstaff, the mechanism of action by which ivermectin targets SARS-CoV-2 is unknown; however, it is thought to be similar to how it works on other viruses – preventing the virus dampening down the host cells’ ability to clear it.
Dr Wagstaff worked with Professor David Jans, also an author on the paper, to conduct the research. Professor Jans and his team have been researching ivermectin’s antiviral properties for more than 10 years with different viruses. The pair said they began their work on SARS-CoV-2 as soon as the pandemic was reported to have started.
Dr Wagstaff concluded by cautioning that the use of ivermectin to combat the COVID-19 pandemic will depend on further pre-clinical and clinical testing which will require funding.
Related topics
Cell Cultures, Disease Research, Drug Leads, Drug Repurposing, RNAs, Therapeutics
Related conditions
Coronavirus, Covid-19, parasites
Related organisations
Monash University's Biomedicine Discovery Institute (BDI), Peter Doherty Institute of Infection and Immunity (Doherty Institute)
Related people
Dr Kylie Wagstaff, Professor David Jans




Where, and how can I buy this?
Looks promising. Here’s to finding funding!
M
The concentration of ivermectin required (ie 2-5 uM) is about 20-50 times the concentration achievable clinically. The dose of the drug can likely be escalated but unlikely to these levels
Actually the human doasage range for IVERMECTIN is 200mcg/kg. So this is very possible! I PRAY they can get funding for more trials!
Actually the peak plasma concentration with that dose is about 70-90 ng/ml (see pk section of FDA label). A 1 uM concentration translates to 875 ng/ml (based on MW of 875). So getting to 2-5 nM as required by the paper is highly unlikely — even more so when one adds the additional complexity of protein binding (ivermectin is highly protein bound 95-99). So free drug concentrations are unlikely to be even close what might be required.
Correction 2-5uM required by the paper. ….
Victor, what about IV infusion to achieve the required concentration in stead of oral form to achieve this plasma concentration?
The issue is weather it can be tolerated at that concentration. Could also consider nebulized drug directly into lungs.
Fight against COVID19
To fight against COVID19 India has to chalk out her own plan; we should not ape other nations. There would be serious socio-economic disaster in India if we continue total or partial lockdown. Most of the COVID19 infected patients in India are asymptomatic; and this might be due to presence of Curcumin in our daily food. So, it is very difficult to determine patient- load in India; we can not test for COVID19 in all persons under the Sun.
Asymptomatic persons would refuse testing and any form of treatment – they would roam freely in society; and would infect the population at large; and this will in near future usher in HERD IMMUNITY in India. Most of the symptomatic patients present with mild to moderate illness; we should treat them as OPD patients. We have to treat these patients with mild-to-moderate Flu-like symptoms as quickly as possible; so that, they do not become severely ill to overwhelm the scarce indoor facilities in our country. Here is a safe outdoor treatment plan that will show quick and effective response in symptomatic patients (for adults): – Vitamin D & C and Zinc daily in usual recommend doses + Virostatic drug Ivermectin 12mg daily for 3 to 5 days + Senolytic antibiotic Azithromycin 250mg to 500mg daily for 5 days to 10 days. This treatment plan ushers in prompt response. Serious indoor patients would get these medicines + necessary supportive measures + Hydroxychloroquine (HCQS). – Arun Kumar Laha 29, Abinash Banerjee Lane. Howrah 711104. ([email protected])
Disclaimer: –
This is not a prescription for general public to self- medicate themselves. It is for medical fraternity to think about my humble suggestion.
To solve the problems of COVID 19, lockdown is not the only way out; we have to rely on development of HERD IMMUNITY by exposing people to low grade infection with COVID19 and we have to treat the symptomatic patients as fast as possible, to cut short the number of serious patients requiring indoor treatment.
Many health- care personnel and other fighters against COVID19 are losing their lives to save the humanity at large; ICMR should think about prescribing IVERMECTIN (12mg in empty stomach once in a week instead of Hydroxychloroquine) to these persons. Ivermectin is safer than Hydroxychloroquine.
Very important that the absorbtion rate is taken into consideration of dosing
We have option using nebulizer for sending ivermectin to lung for fulfilling doze target fotr destriying covit viruses
VERY ENCOURAGING . HOPE IT WORKS INVIVO
good news
If its effectiveness against other viruses ((HIV), Dengue fever, influenza and Zika ) had been established in vitro as early as 2012, why has it not yet been tested as an antiviral in vivo?
Costly medicine will earn more profit for you……Companies all over the world are rushing towards vaccines. Which market has no limit and generation after generation profit…..Think Big so plan Big
Formic Acid, Aspartic Acid, & Glutamic Acid Can Destroy Coronavirus
Ivermectin is a chloride channel agonist, which exist on the surface of brain cells but Ivermectin cannot cross the blood-brain barrier; however, intranasal delivery can deliver drugs to the brain that by-passes the BBB and so the potential for brain effects via this route cannot be excluded.
It is imperative that this Ivermectin is thoroughly tested and used whilst waiting for the vaccine. Hospitals may soon only be able to admit patients who have kept to the social distancing rules, that is outside of 2 metres radially from unrelated public!
Has Ivermectin at 200ug/kg been used in human trial, as in vitro looks very promising. The side effects are well known since it has been used for years in animals and humans globally.
Dose of Ivermectin in Covid-19 is 18mg for 2 days
COVID19 and Ivermectin
High population density plus unlocking lead to community transmission, resulting in fast increase in the number of COVID19 cases in India. Good news is that, large number of infected persons would be cured of the disease without much symptoms; these asymptomatic persons would not subject themselves to any form of testing or treatment; they would roam freely in society and would infect more persons – and this would usher in HERD IMMUNITY in India.
Patients with mild-to-moderate symptoms do not require costly indoor treatment; and such patients should be treated by doctors at the periphery; and the doctors should treat these patients based on clinical assessments of the patients – elaborate testing, to exclude COVID19, for each and every persons suffering from fever with mild-to-moderate Flu-like symptoms is a very costly proposition. There would be severe forms of chaos if we try to cater indoor treatment to all symptomatic patients presenting with mild-to-moderate Flu-like features. However, severely ill patients, aged patients with comorbidities should be admitted in well-equipped institutions.
ICMR should allow doctors, not attached to any state-run hospital or private institution, to treat patients with mild-to-moderate Flu-like symptoms with a cocktail of drugs – Vitamin D, Vitamin C, Zinc, Azithromycin and Ivermectin. Doctors attached to state-run or private institutions are, I think, already instructed by ICMR about management of such patients at indoor and outdoor.
COVID19 is a Global emergency; we do not have much time for elaborate clinical trials (multi-centric, double-blind, placebo-controlled, above all, reports should be acceptable to peer-group-reviewed international Journals) to prove or disprove efficacy of Ivermectin in treating COVID19. Doctors of many nations have published reports proving excellent efficacy of Ivermectin; no doubt, these studies have been conducted with a small number of patients; but still, we cannot discard the inferences of these reports out right, because these findings are collected by senior physicians with long experiences in clinical medicine.
Arun Kumar Laha 09 06 2020
IVERMECTIN
Times Of India published two reports on 21st and 22th May praising Rendesivir and Favipiravir, two antivirals. Mainstream media worldwide are promoting these drugs of big pharma companies; however, most, if not all, antivirals are of questionable efficacy. No one is promoting IVERMECTIN, a cheap oral drug, prescribing which against COVID19 doctors from many nations, including Bangladesh, got EXCELLENT results. No one is interested in promoting this off patented molecule. Scientists could not fabricate vaccines against all and every pathogenic Virus; most probably, COVID19 is one of such type. Monoclonal antibody against viral proteins, however, is a good treatment plan.
NO MASK, NO SOCIAL DISTANCING, NO LOCKDOWN, WILL BE ABLE TO SAVE HUMANITY FROM COVID19. IVERMECTIN, HCQ AND HERD IMMUNITY DUE TO SUBCLINICAL INFECTION IN COMMUNITY AT LARGE WOULD SAVE US.
Fight against COVID19
To fight against COVID19 India has to chalk out her own plan; we should not ape other nations. There would be serious socio-economic disaster in India if we continue total or partial lockdown. Most of the COVID19 infected patients in India are asymptomatic; and this might be due to presence of Curcumin in our daily food. So, it is very difficult to determine patient- load in India; we can not test for COVID19 in all persons under the Sun.
Asymptomatic persons would refuse testing and any form of treatment – they would roam freely in society; and would infect the population at large; and this will in near future usher in HERD IMMUNITY in India. Most of the symptomatic patients present with mild to moderate illness; we should treat them as OPD patients. We have to treat these patients with mild-to-moderate Flu-like symptoms as quickly as possible; so that, they do not become severely ill to overwhelm the scarce indoor facilities in our country. Here is a safe outdoor treatment plan that will show quick and effective response in symptomatic patients (for adults): – Vitamin D & C and Zinc daily in usual recommended doses + Virostatic drug Ivermectin 12mg daily for 3 to 5 days + Senolytic antibiotic Azithromycin 250mg to 500mg daily for 5 days to 10 days. This treatment plan ushers in prompt response. Serious indoor patients would get these medicines + necessary supportive measures + Hydroxychloroquine (HCQS). – Arun Kumar Laha 29, Abinash Banerjee Lane. Howrah 711104. ([email protected])
Disclaimer: –
This is not a prescription for general public to self- medicate themselves. It is for medical fraternity to think about my humble suggestion.
To solve the problems of COVID 19, lockdown is not the only way out; we have to rely on development of HERD IMMUNITY by exposing people to low grade infection with COVID19 and we have to treat the symptomatic patients as fast as possible, to cut short the number of serious patients requiring indoor treatment.
Many health- care personnel and other fighters against COVID19 are losing their lives to save the humanity at large; ICMR should think about prescribing IVERMECTIN (12mg in empty stomach once in a week instead of Hydroxychloroquine) to these persons. Ivermectin is safer than Hydroxychloroquine.