Researchers discover new link between CRISPR, p53 and cancer
Posted: 22 November 2021 | Anna Begley (Drug Target Review) | No comments yet
The new findings suggest that inhibiting p53 can reduce CRISPR-associated cancer risks and may lead to developments in precision medicine.
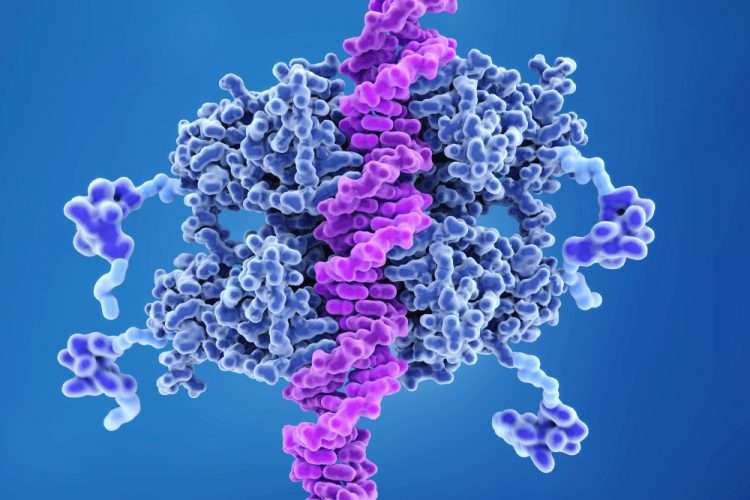

A team at the Karolinska Institutet, Sweden, have identified new links between CRISPR, p53 and other cancer genes that could prevent the accumulation of mutated cells without compromising the gene scissors’ effectiveness, representing a significant step in precision medicine.
NEWS: UTX gene mutation “robs” individuals of natural cancer protection
READ HERE
Previous studies have established that CRISPR is less effective when p53 is active. However, a lack of p53 can allow cells to start growing uncontrollably and become cancerous. In over half of all cancers, the gene for p53 is mutated and thus unable to protect against uncontrolled cell division. It is therefore important to avoid the accumulation of such mutated cells.
This study, published in Cancer Research, used CRISPR screening experiments on isolated cells and analyses of the DepMap database to show that cells with inactivating mutations of the p53 gene gain a survival advantage when subjected to CRISPR and can thus accumulate in a mixed cell population. The researchers have also identified a network of linked genes with mutations that have a similar effect to p53 mutations, and shown that the transient inhibition of p53 is a possible pharmaceutical strategy for preventing the enrichment of cells with such mutations.
“It can seem contradictory to inhibit p53 in a CRISPR context,” commented first author Dr Long Jiang. “However, some of the literature supports the idea that p53 inhibition can make CRISPR more effective. In our study we show that this can also counteract the enrichment of cells with mutations in p53 and a group of associated genes.”
According to the team, the study has a contributory potential to the future clinical implementation of CRISPR in having identified a network of possible candidate genes that should be carefully controlled for mutations when cells are subjected to the CRISPR technique. Another possible conclusion is that the transient inhibition of p53 could prove a strategy for reducing the enrichment of mutated cells.
NEWS: Tropical frog models reveal novel kidney disease insights
READ HERE
The next step of the research is to understand how relevant the described mechanisms are. “In cell cultures, we see a rapid and pronounced enrichment of cells with p53 mutations when we subject the cells to CRISPR, provided, however, that cells with mutations are there from the start,” explained last author Dr Fredrik Wermeling. “So we can show that the mechanism exists and factors that affect it, but do not currently know at what level this is a genuine problem, and that is something we want to explore further in more clinic-centred tests.”
Related topics
CRISPR, Gene Therapy, Genomics, Oncology, Precision Medicine, Small Molecules
Related conditions
Cancer
Related organisations
Karolinska Instituet
Related people
Dr Fredrik Wermeling, Dr Long Jiang