Squaring the circle – cancer vaccines
Posted: 8 May 2019 | Matthias Giese | No comments yet
By the 1890s William B. Coley had injected streptococcal organisms in patients with solid tumours (“Coley’s Toxins”) to activate the immune system. Coley (1862-1936) was an American bone surgeon and pioneer of cancer immunotherapy. He was convinced that post-surgical infections had helped patients to recover better from their cancer by provoking an immune response.
Because of severe adverse effects due to the living streptococcal organisms, he switched to using dead bacteria. But Coley’s published results were difficult to interpret with confidence. “More research would be needed to determine what benefit, if any, this therapy might have for people with cancer” (American Cancer Society). Nevertheless, Coley is known as the “Father of Immunotherapy”.1
Cancer antigens
While Coley’s approach was like flying blind without instruments, modern cancer vaccines are mainly directed against a specific tumour antigen. In principle, every abnormally expressed protein in cancer cells can serve as cancer antigen (CA), based on the recognition by T cells. Since the discovery of the first CA in 1991, MAGE-1 of melanoma cells, a huge plethora of CAs has been described.2 Today, more than 2,000 cancer antigens are identified. The crux of using common CAs is that most of all known cancer antigens are normal but overexpressed cell antigens. Any vaccine against common CAs automatically targets normal cells. In 2009 the National Cancer Institute/ USA published a project on prioritisation of cancer antigens and defined criteria for the “ideal” cancer antigen.3 The ranking criteria were based on the likelihood of efficacy in clinical trials: Therapeutic function > Immunogenicity > Oncogenicity > Specificity > Expression level > Stem cell expression > Number of patient with antigen-positive cancer > Number of antigenic epitopes > Cellular localisation of antigen expression.
Neoantigens
Recent scientific discoveries of patient-specific tumour antigens, neoantigens (present in cancer but not in normal cells) are giving the old idea of cancer vaccines new life. Is selecting a protein target, neoantigen or CA, the key to generate a powerful anti-cancer immune response? Figure 1 illustrates the development of personalised cancer vaccines (P-vaccines) based on patient-specific neoantigens.4 Without question therapeutical cancer vaccines are hot, especially RNA-based vaccines. But do they also work and will they cure the patient?
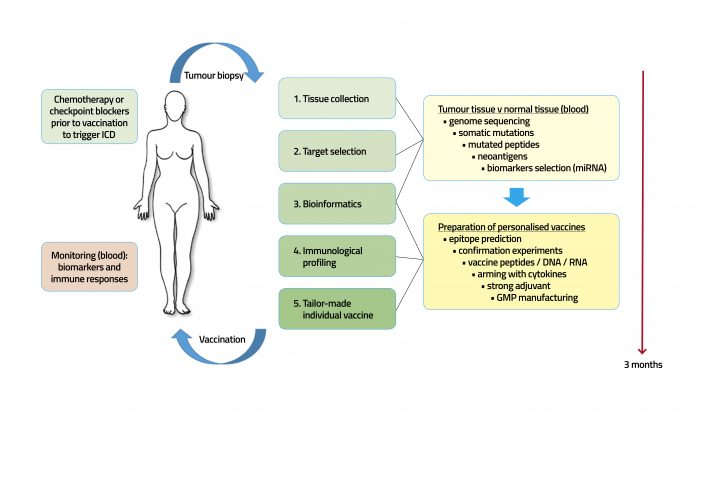
Figure 1:P-vaccine against cancer. The vaccine development begins with an immunological profiling. The aim of individual profiling is to understand the patient-specific interplay between disease and immune system. Tumour-tissue is compared to normal tissue, and targets are identified to tailor a vaccine fitting with measured individual biological conditions. A chemo or immunotherapy with antibodies as checkpoint blockers prior to vaccination can trigger the development of immunogenic cell death (ICD) and prime the immune system with released neoantigens among them the vaccine target antigen should be. Neoantigens additionally stimulate an immune response and thus support checkpoint blockade drugs. The vaccine induced immune response is monitored by the antibody titer and cytotoxic T-cell (CTL) response and specific biomarkers.
An RNA vaccine bears the genetic code for an antigen which is directly translated into protein. RNA vaccines are able to stimulate a complete, humoural and cellular immune response. The same is also true for DNA, or vector driven vaccines or virus-like particles (VLP). However, RNA vaccines are in vogue but nobody is able to explain why they should be superior. The current hype of naked RNA vaccines is the consequence of a successful marketing strategy, not the consequence of clinical performance. The interest of large pharmaceutical companies in this technology unmasks also the lack of innovative own products in this field.
Immune surveillance
The immune system is able to control, at least in part, growth of tumours. In 1863 Rudolf Virchow discovered active leucocytes in tumour tissues. And it was Paul Ehrlich in 1909 who postulated a ‘body’s own protection system’ against tumour cells. ‘The immune surveillance hypothesis’ was born and elaborated by Thomas and Burnet in the 1950s and ‘60s: T-cell-mediated immunity evolved as a specific defence against cancer cells and that T-cells constantly patrol the body, searching for abnormal body cells. As long as the tumour load is controlled by the immune system, as long as this balance between diseases and innate and acquired immunity works, then life is not threatened.
Every day abnormal cells are produced somewhere in the body, are recognised by the immune system and eliminated. Whether this elimination is perfect or imperfect is not clear.
Tumour microenvironment
Probably the immune surveillance leads to a balance between normal and abnormal cells. Pre-cancerous cells stimulate an immune response and are destroyed. But if the immune system can’t detect every malignant cell, and at the same time a suitable tumour microenvironment (TME) is formed by inflammatory processes, carcinogenesis is no longer blocked.4 The dual functions of immune cells arise in TME. While outside the TME T-cells, B cells, macrophages etc protect the body against invaders, within TME these cells contribute to an immunosuppressive milieu. All solid tumours contain infiltrates of leukocytes – among these are T-cells (Th1, Th2, Th17, Treg, γδ T cells, CTLs) and tumour-associated macrophages (TAMs). Immune cells now stimulate the tumour proliferation and angiogenesis.
Immunoediting
Within the TME the immune system has successfully edited these cancer cells and the cancer cells have their “lessons learned”. It is not surprising: the evolutionary mandate of life means to adapt to the environment; to permanently improve the genetic configuration – everything to do for survival only. The MDR (multidrug resistance) pump system in many cancer cells underlines this survival strategy, and immunoediting is only another mechanism of the evolutionary survival strategy. The mechanisms leading to immune tolerance against malignant cells are unknown.
A therapeutical cancer vaccine is confronted with several massive natural barriers: the efficacy is limited by the local tolerance of T-cells within the immunosuppressive milieu (TME). Tumour antigens are often “self” proteins or masked antigens or weak neoantigens. Overall weakness of the immune system (central immunosenescence) due to chronic inflammation accompanying most cancer diseases. Also, ineffective adjuvants contribute to this failure. Ageing has a very deep impact an cancer vaccines.
The secret
The key to generating a powerful anti-cancer immune response is not the accurate antigen; whether in the form of RNA, DNA, vectors or VLPs. The only key to developing powerful therapeutical cancer vaccines is first to decode the secrets of the inbalanced immune system and second to reconstitute it. The correlation of immune dysfunctions and the development of tumours is evident.5 There is no sense in creating any cancer vaccine while ignoring these clear coherences. But this is what currently happens! Behind the hype of RNA cancer vaccines is the same boring attitude as in the last two decades: Take an antigen, put this antigen in a suitable packing (or not), look for an appropiate delivery system and bye, bye. And this construct shall work? It did not work in the past and it will not work today or in the future. Old wine in new wineskins. What a giant RNA bubble is about to explode.
The first RNA cancer vaccine
CureVac Prostate Cancer Vaccine Candidate fails Phase IIb Trial (2017): CureVac’s lead mRNA drug was the first RNA vaccine in man. “These results…pave the way for us to advance more potent prophylactic vaccine formulations into the clinic,” said Ingmar Hoerr, CEO of CureVac.6 To be honest, the world is not waiting for another rabies vaccine but for the first therapeutical cancer vaccine. In 2019, CureVac celebrates its 19th anniversary: no cancer vaccine on the market. BioNTech, founded in 2008, has engaged 700 employes: no cancer vaccine on the market. Manpower is not enough. Worldwide, there are no successful clinical trials with RNA cancer vaccines, regardless of the providing company.
Conclusion
Immunotherapy by vaccination is possible, based on the reconstitution of immune functions. The Immunosenescence, immune dysfunctions are not limited to the process of ageing but also linked to cancer diseases. It makes no sense at all to vaccinate a cancer patient suffering from loss of important immune functions. A risk analysis before vaccination could clear the current disposition of the immune system and open a possible therapeutical window. Would this make sense for a therapeutical vaccination or not?4
Biography
Matthias Giese has studied Molecular Biology and Biochemistry. He got a first PhD in Tumour Immunology and a major PhD (venia legendi) in Molecular Virology. His industrial team in Vienna started the first European multicentre clinical trial with a personalised anti-melanoma vaccine. Matthias also developed the first successful therapeutic DNA vaccine against a chronic viral infection in horses. Currently he is working as an independent consultant (IMV Heidelberg). His focus is on neoantigenes, therapeutical vaccines, and the interaction with immune checkpoint modifiers. Matthias is the author of the textbook “Introduction to Molecular Vaccinology“ and other specialised books on modern vaccines.
References
- McCarthy EF. The Toxins of William B. Coley and the Treatment of Bone and Soft-Tissue Sarcomas. Iowa Orthop J.2006; 26: 154–8.
- van der Bruggen, Traversari C, Chomez P, Lurquin C, De Plaen E, van den Eynde B, et al. A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science 1991 Dec; 254: 1643-7.
- Cheever MA, Allison JP, Ferris AS, Finn OJ, Hastings BM, Hecht TT, et al. The prioritization of cancer antigens: a national cancer institute pilot project for the acceleration of translational research. Clin Cancer Res. 2009 Sep; 1;15(17):5323-37.
- Giese M. Introduction to Molecular Vaccinology. (Textbook). 1st ed. Heidelberg/ New York: Springer; 2016.
- Ostrand-Rosenberg S. Immune surveillance: a balance between protumor and antitumor immunity. Curr Opin Genet Dev. 2008 Feb; 18(1):11-8.
- org [Internet]. Washington: CureVac’s lead mRNA drug is down but not out, says CEO; [cited Jan 12, 2017]. Available from: https://www.eatright.org/.https://www.fiercebiotech.com/biotech/curevac-s-lead-mrna-drug-down-but-not-out-says-ceo;
The rest of this content is restricted - login or subscribe free to access
 Thank you for visiting our website. To access this content in full you'll need to login. It's completely free to subscribe, and in less than a minute you can continue reading. If you've already subscribed, great - just login.
Thank you for visiting our website. To access this content in full you'll need to login. It's completely free to subscribe, and in less than a minute you can continue reading. If you've already subscribed, great - just login.
Why subscribe? Join our growing community of thousands of industry professionals and gain access to:
- quarterly issues in print and/or digital format
- case studies, whitepapers, webinars and industry-leading content
- breaking news and features
- our extensive online archive of thousands of articles and years of past issues
- ...And it's all free!
Click here to Subscribe today Login here
Related topics
Antibodies, Drug Discovery, Vaccine
Related conditions
Cancer
Related people
William B. Coley







