Out of the US, a newly developed a rapid, stain-free, and automated viral plaque detection system enabled by holography and deep learning, is a cost-effective way of observing the formation of viral plaques caused by viral infections over a layer of cells.
Viral infections have challenged humanity for centuries. Even with progressive scientific advancements, the struggle against viruses continues, as exemplified by the recent COVID-19 pandemic. In the fight against these viral infections, a variety of techniques have been established for detecting and quantifying viruses, contributing significantly to the development of critical vaccines and antiviral medications.
Among these techniques, the viral plaque assay stands out as the gold standard due to its unique ability to assess virus infectivity in a cost-effective way by observing the formation of viral plaques caused by viral infections over a layer of cells.
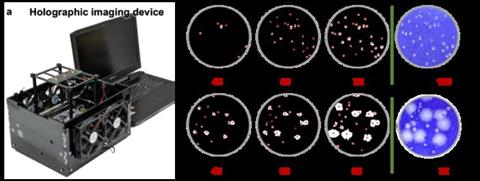
In a new paper published in Nature Biomedical Engineering, a team of scientists led by Professor Aydogan Ozcan from the Electrical and Computer Engineering Department at UCLA and an associate director of the California NanoSystems Institute, US, have developed a rapid, stain-free, and automated viral plaque detection system enabled by holography and deep learning. This system incorporates a cost-effective and high-throughput holographic imaging device that continuously monitors the unstained virus-infected cells during their incubation process. At each imaging cycle, these time-lapse holograms captured by the device are periodically analysed by an AI-powered algorithm to automatically detect and count the viral plaques that appear due to virus replication.
Nevertheless, the traditional viral plaque assays require an incubation period of 2~14 days, followed by sample staining using chemicals and human visual inspection to count the number of viral plaques. This procedure is time-consuming and susceptible to staining artifacts and counting errors induced by human technicians. Therefore, an accurate, automated, rapid, and cost-effective viral plaque quantification technique is urgently needed.

Viral plaque detection system enabled by holography and deep learning (Credit: Ozcan Lab)[/caption]
The proof-of-concept and effectiveness of the new system were demonstrated using three different types of viruses: vesicular stomatitis virus (VSV), herpes simplex virus type-1 (HSV-1), and encephalomyocarditis virus (EMCV). By utilising this system, UCLA researchers achieved the detection of over 90 percent of VSV viral plaques within 20 hours of incubation without any chemical staining, demonstrating a time saving of more than 24 hours in comparison to the traditional plaque assay, which requires 48 hours of sample incubation. In the case of HSV-1 and EMCV, this system effectively reduced their viral plaque detection times by approximately 48 and 20 hours, respectively, compared to the detection time needed for the traditional staining-based viral plaque assay.
In addition to offering major time savings, this stain-free and cost-effective system can successfully identify individual viral plaques within clusters as opposed to the traditional viral plaque assays, which fail to separately detect and count those individual plaques within clusters due to the spatial overlap of their signatures.