Genome-edited iPSCs show promise for treating Fabry Disease
Posted: 4 July 2023 | Izzy Wood (Drug Target Review) | No comments yet
Japanese researchers successfully engineered iPSCs to secrete a modified enzyme, mNAGA, and restored enzyme activity in vitro and in a mouse model, opening new avenues for regenerative medicine for conditions such as Fabry Disease.

Researchers have made significant strides in the field of regenerative medicine with the use of induced pluripotent stem cells (iPSCs). These cells, derived from healthy individuals, have shown great potential in various clinical trials. However, a recent study suggests that the transplantation therapy using iPSCs can be further enhanced through genetic modification.
In a groundbreaking proof-of-concept experiment, scientists from Tokyo Metropolitan Institute of Medical Science, Japan, a mouse model of Fabry disease to explore the possibilities of genetic modification in iPSCs. Fabry disease is a genetic disorder caused by the deficiency of an enzyme called α-Galactosidase A (GLA), which leads to the accumulation of harmful substances in the body. To address this issue, researchers had previously developed a modified enzyme, known as modified α-N-acetylgalactosaminidase (mNAGA), which showed promise in curing Fabry disease.
The team embarked on this study to investigate whether iPSCs, engineered to secrete mNAGA through genome editing, could provide the missing GLA activity in vivo. To achieve this, they employed TALEN-mediated knock-in techniques to introduce mNAGA secretion into the iPSCs at a safe harbor locus called the AAVS1 site. In addition, they used CRISPR-Cas9 to disrupt the endogenous GLA gene in the iPSCs, aiming to eliminate possible immunogenic reactions in patients.
In vitro experiments involving co-culturing of the engineered iPSCs with Fabry disease model cardiomyocytes and fibroblasts revealed promising results. The mNAGA-expressing iPSCs successfully restored the GLA activity in these cells, effectively addressing the deficiency.
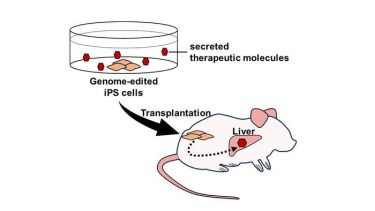

Genome editing enables the expression and secretion of therapeutic molecules from iPSCs. Furthermore, transplantation of these cells allows for the delivery of therapeutic molecules to organs and tissues in vivo. (Credit: TMIMS)
Moving forward, the team conducted in vivo experiments by transplanting the iPSCs secreting mNAGA into the testes of Fabry disease model mice. After 7 to 8 weeks, a significant improvement in GLA activity was observed in the liver, while no recovery was observed in the heart, kidney, or blood plasma. Although the levels of harmful substances in the liver, known as globotriaosylceramide (Gb3) and globotriaosylsphingosine (Lyso-Gb3), were not reduced, it was determined that the limited amount of mNAGA secreted by the transplanted iPSCs was insufficient to achieve this effect.
Despite the current limitations, the study highlights the potential for further advancements. Researchers believe that enhancing the amount of secreted mNAGA through genome editing techniques may be a possibility in the future. Additionally, they propose the direct delivery of mNAGA to specific organs and tissues in need of GLA activity. For example, transplanting a cardiomyocyte sheet derived from iPSCs secreting mNAGA could directly deliver the therapeutic molecule to the heart. Moreover, the study indicates that this strategy can be applied to other diseases beyond Fabry disease.
This research serves as a testament to the potential of cell therapy using genome-edited iPSCs that secrete therapeutic molecules. These engineered iPSCs not only offer a resource for cell transplantation but also serve as a promising drug delivery system. Although further studies and advancements are necessary, these findings pave the way for future breakthroughs in regenerative medicine and offer hope for patients with genetic disorders like Fabry disease.
Related topics
Animal Models, Genome Editing, In Vitro, In Vivo, Induced Pluripotent Stem Cells (iPSCs)
Related conditions
Fabry Disease
Related organisations
Tokyo Metropolitan Institute of Medical Science